Receiving a cancer diagnosis is a life-altering event that often triggers an immediate, overwhelming need for information. Among the terminology that patients and their families frequently encounter is the "clinical trial." While the phrase is common, its implications, opportunities, and realities are often misunderstood. Far from being a "last resort," clinical trials represent the engine of medical progress—the bridge between laboratory discovery and the standard of care that saves lives every day.
The Evolution of Cancer Care: Why Clinical Trials Matter
At their core, cancer clinical trials are rigorous research studies designed to evaluate new ways to prevent, detect, or treat malignancy. They are the bedrock of oncology; every treatment currently recognized as the "standard of care"—from foundational chemotherapy to cutting-edge immunotherapies—was once rigorously tested in a clinical trial.
The scope of these trials is vast. While many focus on testing novel therapeutic agents, others are designed to improve early screening techniques, refine diagnostic precision, or enhance the quality of life for patients by mitigating the debilitating side effects of traditional treatments. By participating in a trial, patients aren’t just accessing the latest medical science; they are contributing to a global body of knowledge that will improve outcomes for future generations.
As Dr. Joshua Brody of the Icahn School of Medicine at Mount Sinai aptly notes, "Every miraculous breakthrough has come from clinical trials."
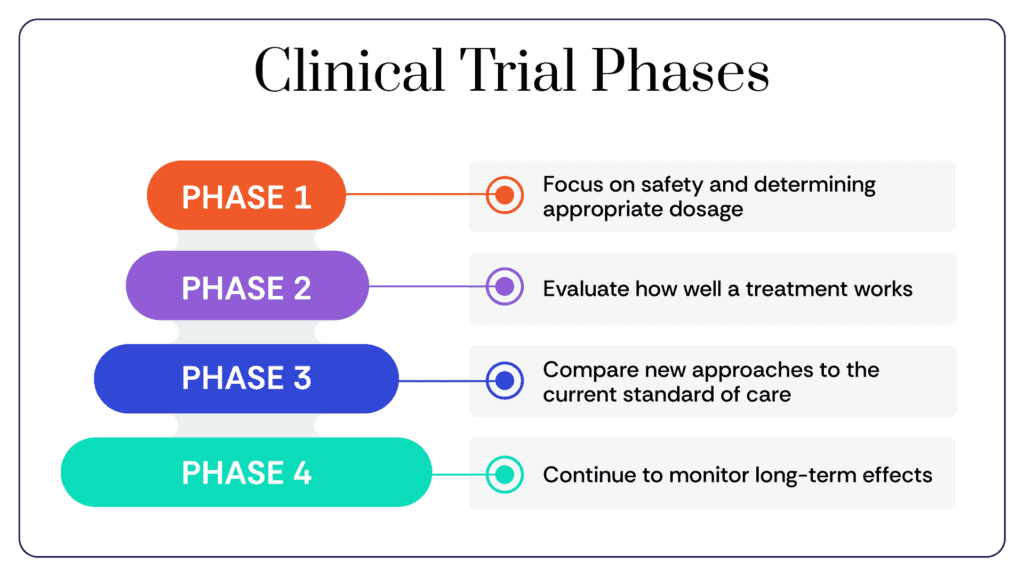
The Chronology of Discovery: Understanding the Phases
The path from a scientific hypothesis to an FDA-approved treatment is a deliberate, multi-stage journey. This structure is designed to prioritize patient safety while gathering essential data on efficacy.

Phase I: The Foundation of Safety
The primary objective of a Phase I trial is safety. Researchers determine the best dose of a new treatment, how it should be administered, and how the human body reacts to it. These trials typically involve a small group of participants and are critical for identifying potential toxicities early.
Phase II: Measuring Efficacy
Once a treatment has been deemed safe, Phase II trials begin to assess whether it actually works against specific types of cancer. Researchers look for evidence of tumor shrinkage or improvements in biomarkers, effectively "testing the proof of concept" on a larger cohort of patients.
Phase III: The Gold Standard
Phase III trials are the most definitive. They compare the new treatment against the current standard of care. By enrolling hundreds or even thousands of patients, these trials provide the statistical power necessary to prove that the new intervention is superior or equivalent to existing options.
Phase IV: Post-Marketing Surveillance
After a drug receives regulatory approval, Phase IV trials continue to monitor its performance in the broader population. This stage helps identify long-term side effects or rare adverse reactions that might not have appeared in smaller, shorter-term studies.
Debunking the Myths: When is the Right Time?
One of the most persistent misconceptions in oncology is that clinical trials are only for those who have exhausted all other options. This could not be further from the truth. Clinical trials are available at almost every stage of the treatment journey—from initial diagnosis to recurrence.

For some, a trial might offer a more targeted, personalized approach early on, potentially sparing the body from the systemic toll of traditional chemotherapy. For others, it provides a vital lifeline when standard protocols have proven ineffective. The decision to enter a trial is deeply personal and requires an honest, ongoing dialogue with a medical team.
Supporting Data: The Patient Perspective
The impact of these trials is best illustrated by those who have walked the path. Kristin Kleinhofer, a Stage 4 Leukemia survivor, emphasizes the dual nature of the experience: "I began to look at clinical trials, and it wasn’t just that I was fighting for my life, but I really wanted to pay it forward, help cancer research, and help other cancer patients, even if it wasn’t going to work for me."
Sharon Belvin, a survivor of Stage 4 Melanoma, echoes this sentiment: "Clinical trials are the lifeline for long-term survival rates with cancer. There is no other way to move this field forward. If it wasn’t for clinical trials, I wouldn’t be here."
These testimonials underscore that while trials are a professional, data-driven endeavor, they are deeply human experiences. Participation offers patients closer monitoring by specialized care teams, providing an extra layer of support and observation throughout the treatment cycle.
Practical Considerations: What Patients Need to Know
Entering a clinical trial is a significant decision that requires weighing potential benefits against personal constraints.

Eligibility Criteria
Not every trial is a match for every patient. Researchers establish strict eligibility criteria based on tumor biology, genetic markers, prior treatment history, and overall physical health. These criteria are not meant to exclude, but to ensure that the study can produce accurate, safe, and meaningful results.
The Financial Aspect
A common concern for patients is the cost of care. In the majority of clinical trials, the study sponsor covers the cost of the experimental treatment and any procedures required specifically by the trial protocol. However, "routine care"—the standard doctor visits and tests that would occur regardless of the trial—are often billed to the patient’s insurance. It is imperative that patients verify coverage with their providers before enrolling.
Lifestyle and Logistics
Clinical trials often require a higher frequency of clinic visits, additional blood work, and more rigorous monitoring. Patients must consider the logistics: How far is the treatment center? Do I have a support system for transportation? Can I balance this schedule with my professional and family life?
Empowering the Conversation: 10 Questions to Ask Your Doctor
To navigate the landscape of clinical trials effectively, patients should go into consultations prepared. The following questions can help turn a complex medical topic into a clear, actionable plan:
- Are there any clinical trials that might be right for me? Inquire about specific studies based on your diagnosis, stage, and genetic markers.
- What is the primary goal of this trial? Understand whether the focus is on curing, managing symptoms, or improving quality of life.
- How does this compare to my current options? Ask your doctor to explain why this specific trial might be a superior or necessary alternative to standard care.
- What are the potential benefits? Seek a realistic view of what "success" looks like in the context of this study.
- What are the potential risks? Discuss both common and rare side effects, including those that are currently unknown.
- What phase is the trial in? Knowing if it is early-phase or late-phase helps contextualize how much is already known about the therapy.
- What is the time commitment? Ask about the number of clinic visits, hospitalizations, and diagnostic tests required.
- What happens if the treatment fails? Ensure you have a "Plan B"—will you be able to switch to another therapy if the trial does not yield results?
- What are the financial implications? Clearly identify which costs are covered by the sponsor and which remain the responsibility of your insurance or yourself.
- How will this affect my quality of life? Discuss the impact on your daily routine, energy levels, and long-term well-being.
Implications for the Future of Medicine
The medical community is currently in a renaissance of cancer research, with immunotherapy and precision medicine leading the charge. Clinical trials are the mechanism by which these revolutionary ideas are validated. Every patient who steps forward to participate in a trial is a partner in the evolution of medicine.
Navigating this path can feel daunting, but you are not alone. Resources such as the CRI Clinical Trial Finder and professional trial navigators are available to help patients decipher complex information and identify studies that align with their specific medical profiles.
Ultimately, a clinical trial is more than a research study; it is an act of hope. Whether or not it is the right path for your specific situation, the act of asking the question can open doors to new possibilities. By staying informed, asking the right questions, and collaborating closely with your oncology team, you can make a decision that prioritizes your health, your values, and your future.


















