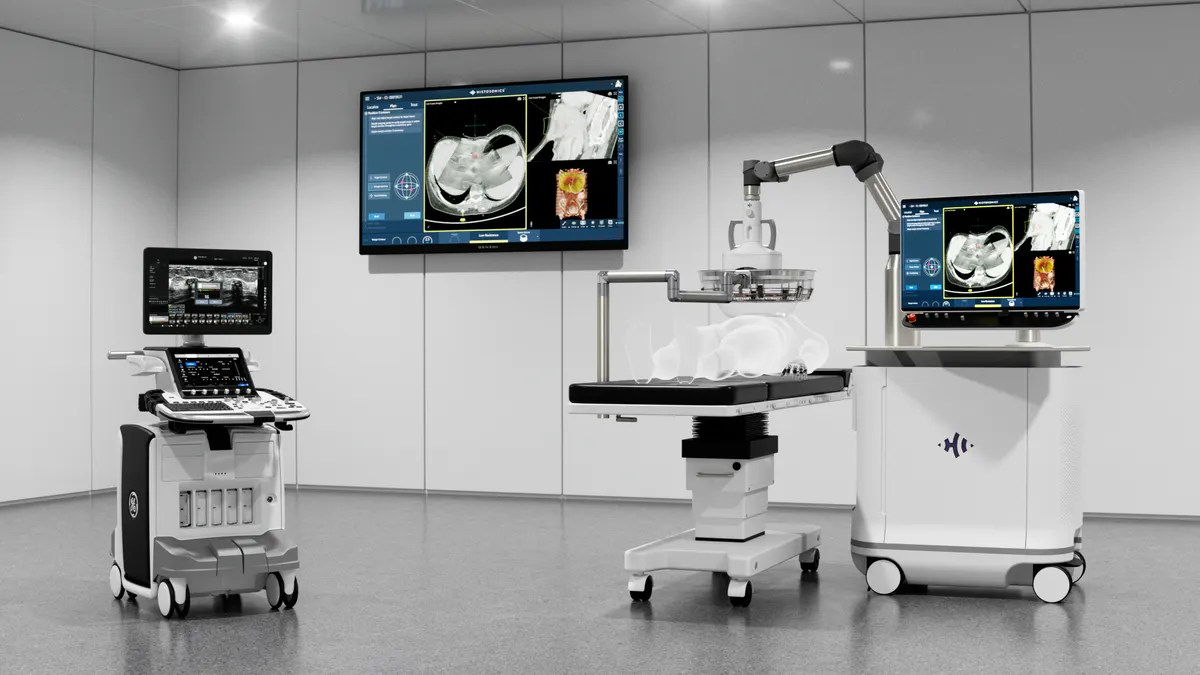
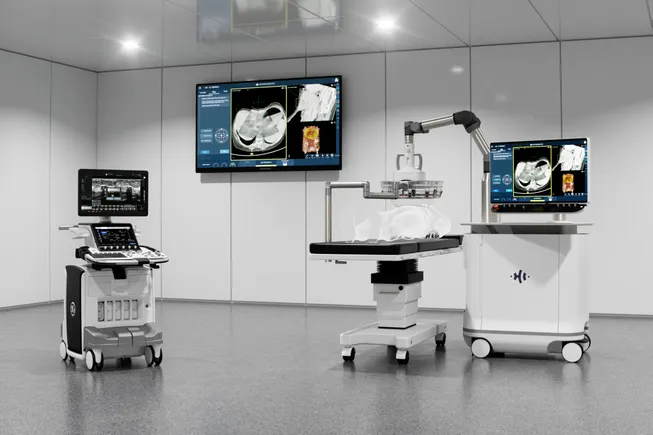
In the rapidly evolving landscape of medical technology, few innovations have captured the attention of both the clinical community and major venture capital firms as effectively as HistoSonics’ Edison platform. By harnessing the power of pulsed sound waves to mechanically destroy targeted tissue, the company is positioning itself at the forefront of a paradigm shift in oncology and beyond. With the backing of high-profile investors and a clear path toward expanding its clinical indications, HistoSonics is transitioning from a high-potential startup to a dominant force in non-invasive surgical intervention.
The Core Technology: Mechanical Precision Through Sound
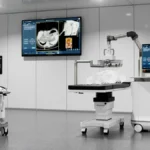
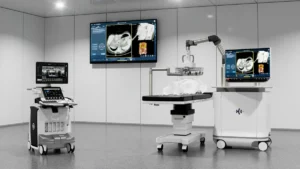
The Edison system operates on a principle known as histotripsy. Unlike traditional surgical methods that rely on incisions, or thermal ablation techniques that utilize heat or cold to destroy tissue, histotripsy is a purely mechanical, non-invasive process.
The system utilizes focused, pulsed sound waves to generate micro-bubbles within a targeted area. These bubbles form and collapse with such extreme precision and rapidity that they physically disrupt the structure of the target cells, effectively liquefying the tumor. Because the process is guided by real-time imaging, clinicians can ensure the destruction of the malignancy while leaving surrounding healthy tissue, blood vessels, and nerves intact.
This approach offers significant advantages over conventional surgical procedures. By eliminating the need for incisions, the Edison system reduces the risk of post-operative complications such as bleeding and infection. Furthermore, because it does not rely on thermal energy, it avoids the common pitfalls of thermal ablation, where unintended heat spread can cause collateral damage to organ function.
A Chronology of Clinical Milestones
HistoSonics’ trajectory has been defined by a methodical, data-driven approach to regulatory approval and clinical validation.
- 2023: The Liver Cancer Breakthrough: A defining moment for the company occurred in 2023, when the U.S. Food and Drug Administration (FDA) granted de novo authorization for the Edison system in the treatment of liver cancer. This approval served as a critical proof-of-concept, demonstrating that the technology could safely and effectively target complex abdominal tumors.
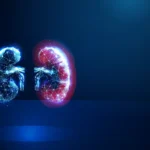
- 2024: Pivotal Kidney Cancer Trials: Building on the success in liver oncology, HistoSonics pivoted toward kidney cancer. The company initiated a pivotal clinical trial designed to evaluate the safety and efficacy of histotripsy for renal tumors. This trial specifically measures the percentage of targeted tumors successfully eliminated after a single session.
- Current Expansion: Beyond these initial focal points, the company has begun exploring the application of the Edison system for pancreatic tumors and benign prostatic hyperplasia (BPH). This expansion represents a broader vision: to establish histotripsy as a standard-of-care modality across a vast array of benign and malignant conditions.
Supporting Data and Market Potential
The commercial viability of HistoSonics is anchored in the sheer scale of the patient populations it seeks to serve. According to estimates from the American Cancer Society, physicians in the United States diagnose approximately 80,000 new cases of kidney cancer annually. When coupled with the 42,000 new liver cancer diagnoses each year, the addressable market for the Edison system is substantial.
However, the value proposition extends beyond market size; it is a matter of clinical necessity. Current treatments for renal tumors—ranging from partial nephrectomy (surgical removal) to thermal ablation—carry inherent risks. Patients undergoing these procedures often face long recovery times and the potential for secondary injury to the affected organ. Histotripsy provides a compelling alternative, promising the "surgical" destruction of a tumor without the physiological trauma of a traditional operating room experience.
HistoSonics’ internal data from its ongoing clinical trials continues to yield promising insights. The focus on single-session elimination is particularly attractive to health systems looking to optimize outpatient throughput and reduce the financial burden associated with multi-day hospital stays and complex recovery protocols.
Financial Backing and Strategic Vision
The confidence of the investment community in HistoSonics’ vision cannot be overstated. Last year, the company secured a landmark $250 million financing round, an infusion of capital aimed at accelerating its expansion into new global markets and broadening its clinical indications.
Notably, the funding round included support from heavyweights such as Bezos Expeditions and Thiel Bio, alongside Wellington Management and K5 Global. In a significant validation of the company’s long-term worth, investors participated in a deal that valued HistoSonics at approximately $2.25 billion. This valuation reflects not just the current capabilities of the Edison system, but the disruptive potential of the underlying histotripsy platform. By securing such significant financial runway, HistoSonics is positioned to sustain long-term research and development efforts, ensuring the platform remains at the cutting edge of medical physics.
Implications for the Future of Surgery
The rise of histotripsy signals a broader trend in medicine: the "dematerialization" of surgery. As technology moves away from scalpels and toward precise, energy-based interventions, the definition of an "operating room" is likely to change.
Improving Patient Outcomes
For the patient, the implications are profound. The shift toward non-invasive procedures minimizes hospital stays and allows for a quicker return to daily life. For elderly patients or those with co-morbidities who may be poor candidates for traditional surgery, histotripsy could represent the difference between life-extending treatment and palliative care.

The Economic Impact on Healthcare
Health systems globally are under immense pressure to reduce costs while improving outcomes. By moving tissue destruction from the operating theater to the interventional suite, HistoSonics potentially reduces the demand on anesthesia services, sterile surgical environments, and post-operative nursing care. As the technology gains more clinical data, it is expected that payers will increasingly view histotripsy as a cost-effective alternative to traditional oncological surgeries.
Challenges Ahead
Despite the optimism, HistoSonics faces the typical hurdles of any disruptive medical device firm. The company must continue to navigate complex regulatory pathways in international markets, each with its own set of requirements. Furthermore, as the Edison system enters more clinical settings, the company must maintain a robust training program for physicians to ensure the technology is used with the precision required for optimal outcomes. The transition from trial-based success to widespread clinical adoption requires not just a superior machine, but a superior implementation strategy.
Conclusion
HistoSonics is at the center of a technological revolution that promises to redefine how we treat some of the most challenging diseases in modern medicine. By blending advanced imaging with the mechanical precision of pulsed sound, the Edison system offers a glimpse into a future where "surgical" outcomes are achieved without the trauma of physical incision.
With the backing of some of the world’s most sophisticated investors and a growing body of evidence supporting its efficacy in liver and kidney cancers, the company is well-poised to expand its footprint. As trials for pancreatic tumors and other conditions progress, the industry will be watching closely to see if histotripsy can indeed become the next great cornerstone of non-invasive oncology. In an era where the focus of medicine is shifting toward minimally invasive and high-precision interventions, HistoSonics is not just participating in the conversation—it is leading it.