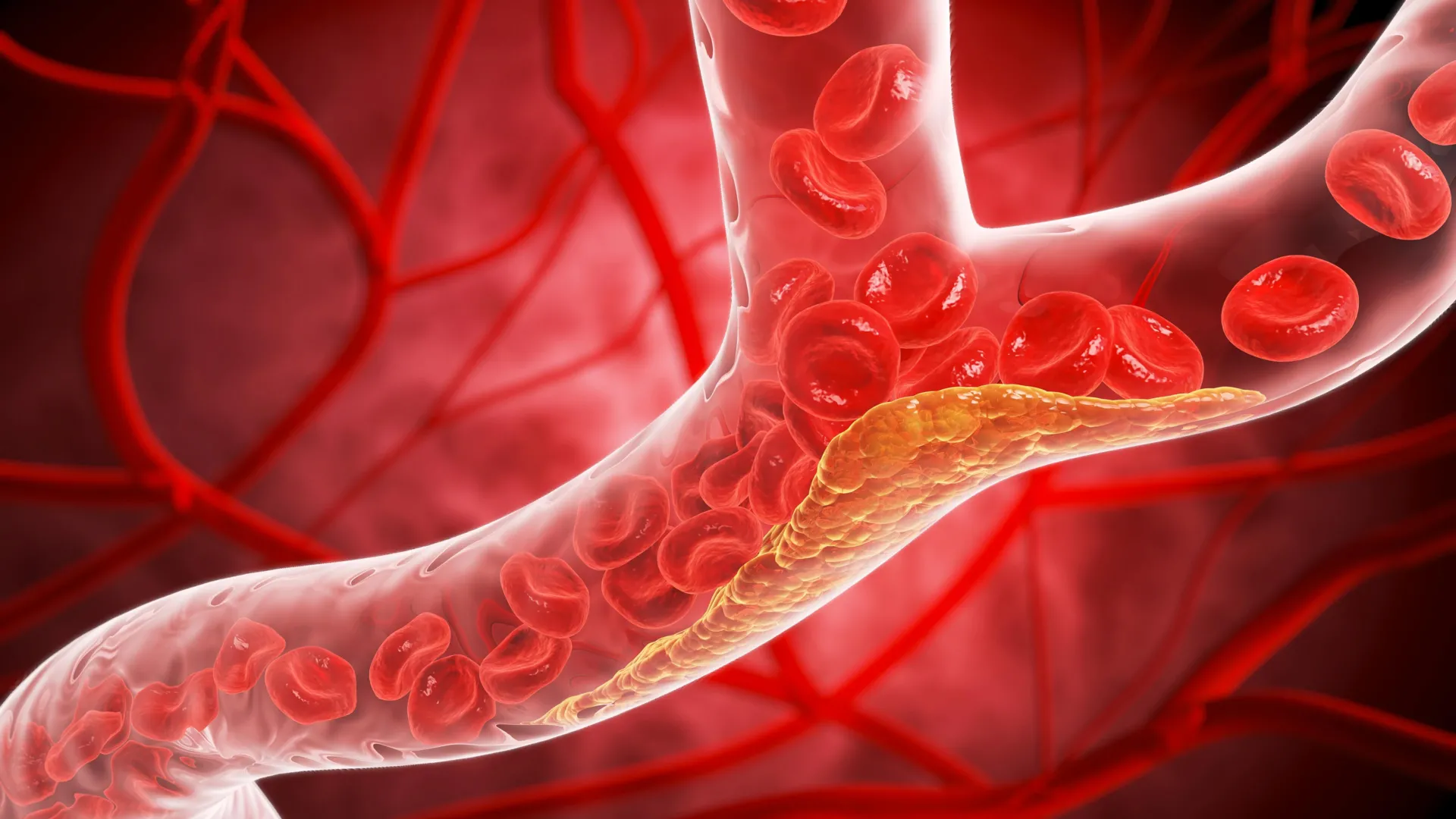
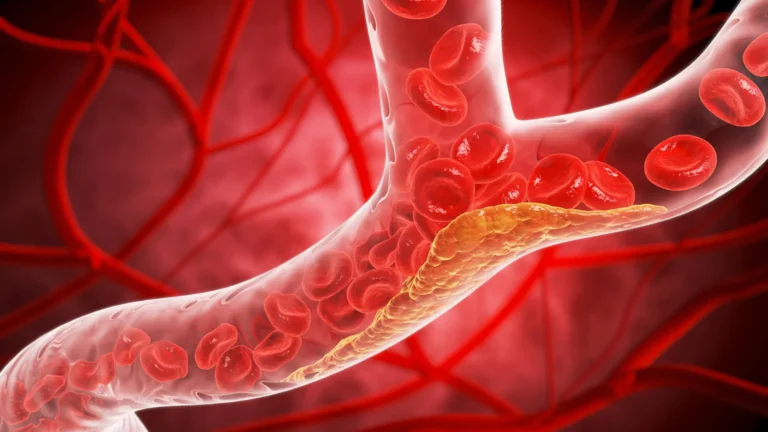
In the landscape of modern cardiovascular medicine, few metrics are as critical—or as difficult to manage—as low-density lipoprotein (LDL) cholesterol. Often colloquially dubbed "bad cholesterol," this waxy substance is the primary culprit in the development of atherosclerosis, the hardening and narrowing of arteries that serves as the precursor to heart attacks and strokes. Despite the availability of potent therapies, a significant gap remains in clinical practice: fewer than half of patients with established atherosclerotic cardiovascular disease successfully reach their cholesterol targets.
That narrative may be on the verge of a historic pivot. A groundbreaking phase three clinical trial, recently published in The New England Journal of Medicine, has introduced an experimental oral medication known as enlicitide. The study indicates that the once-daily pill can reduce LDL cholesterol levels by as much as 60%. Should it secure approval from the U.S. Food and Drug Administration (FDA), enlicitide could represent the most significant advancement in lipid-lowering therapy since the introduction of statins, offering a convenient, high-efficacy solution for millions of Americans at high risk for cardiac events.
The Chronology of a Breakthrough: From Nobel Laureates to Modern Medicine
The development of enlicitide is not an isolated discovery; rather, it is the culmination of decades of rigorous scientific inquiry rooted in the laboratories of UT Southwestern Medical Center (UTSW). The story of lipid management is one of the great success stories of 20th-century medicine, and its current trajectory owes a profound debt to past pioneers.
The Foundation: The LDL Receptor
In the late 20th century, UTSW researchers Michael Brown, M.D., and Joseph Goldstein, M.D., fundamentally changed the world’s understanding of cholesterol. Their discovery of the LDL receptor—the mechanism by which liver cells effectively clear cholesterol from the bloodstream—provided the biological blueprint for modern heart disease treatment. This work earned them the Nobel Prize in Physiology or Medicine in 1985 and directly catalyzed the development of statins, which remain the cornerstone of cholesterol therapy today.
The Genetic Clue: The Dallas Heart Study
Building on this legacy, researchers Helen Hobbs, M.D., and Jonathan Cohen, Ph.D., utilized the landmark Dallas Heart Study to identify a genetic anomaly. They discovered that certain individuals possessed natural, life-long low levels of LDL cholesterol due to a genetic variant that suppressed the production of a protein called PCSK9. This protein acts as a "brake" on the liver’s ability to clear cholesterol; when inhibited, the liver clears LDL far more efficiently.
This revelation led to the creation of injectable PCSK9 inhibitors—monoclonal antibodies and RNA-based therapies—which effectively mimic the genetic protection identified by Drs. Hobbs and Cohen. These injectable drugs, such as evolocumab and alirocumab, proved that reducing PCSK9 activity could slash LDL levels by roughly 60%.
The Oral Challenge
While the injectable inhibitors were a triumph of pharmacology, they faced a hurdle in the real world: patient compliance and accessibility. Because they required injections, many patients were reluctant to use them. Furthermore, high costs and insurance barriers often relegated these drugs to second- or third-line treatments. Enlicitide emerged as the solution to this "delivery problem," successfully translating the biological efficacy of the injectable inhibitors into a daily oral tablet.
Supporting Data: The Phase Three Clinical Trial
To evaluate the safety and efficacy of enlicitide, researchers conducted a massive phase three clinical trial involving 2,909 participants. The cohort consisted entirely of individuals who either had established atherosclerotic cardiovascular disease or were at high risk due to related health conditions.
The "Real World" Participant Profile
A critical strength of this trial, according to lead investigator Ann Marie Navar, M.D., Ph.D., was the composition of the study population. "The study population reflects what we see in clinical practice," Dr. Navar noted. "Even the highest intensity statins are often not enough to get people to their cholesterol goals."
At the start of the study, the vast majority of participants were already on statin therapy. Despite this, their average LDL cholesterol level was 96 mg/dl. For patients with established cardiovascular disease, the recommended clinical goal is often 70 mg/dl, and for those at extreme risk, it is as low as 55 mg/dl. The participants represented the "difficult-to-treat" population that cardiologists encounter daily.
The Results
Participants were randomized into two groups: one receiving the experimental enlicitide pill and the other receiving a placebo. The results after 24 weeks were striking. Those taking enlicitide saw their LDL cholesterol levels drop by approximately 60% compared to the placebo group.
Beyond LDL reduction, the drug demonstrated positive secondary outcomes, effectively lowering other markers associated with cardiovascular risk, including:
- Non-HDL lipoprotein cholesterol
- Apolipoprotein B
- Lipoprotein(a)
Importantly, these improvements were not fleeting; the data confirmed that these reductions were maintained over a full year of follow-up. Dr. Navar, who serves as an Associate Professor of Internal Medicine and in the Peter O’Donnell Jr. School of Public Health at UT Southwestern, emphasized the magnitude of the finding: "These reductions in LDL cholesterol are the most we have ever achieved with an oral drug by far since the development of statins."
Official Responses and Clinical Implications
The medical community has reacted with cautious optimism, viewing enlicitide as a potential "game changer" for preventative cardiology.
Bridging the Gap in Care
Dr. Navar highlighted the persistent struggle physicians face in guiding patients to their targets. "Fewer than half of patients with established atherosclerotic cardiovascular disease currently reach LDL cholesterol goals," she stated. "An oral therapy this effective has the potential to dramatically improve our ability to prevent heart attacks and strokes on a population level."
The primary advantage of enlicitide is its form factor. By shifting from a complex, injectable regimen to a simple daily pill, the burden on the patient is significantly reduced. This ease of use is expected to improve medication adherence—a major hurdle in treating chronic, asymptomatic conditions like high cholesterol.
The Next Frontier: Outcomes Data
While the reduction in LDL cholesterol is statistically undeniable, the medical community is now looking toward the next phase of evidence. A separate clinical trial is currently underway to definitively prove that these lowered cholesterol levels translate directly into a reduction in clinical "events"—specifically, fewer heart attacks and strokes. This outcomes-based data will be the final piece of the puzzle required to fully integrate enlicitide into standard clinical guidelines.
Ethical and Financial Disclosures
The research surrounding enlicitide was sponsored by the pharmaceutical manufacturer Merck & Co. Inc. As is standard in high-impact medical publishing, the researchers involved have disclosed their financial relationships.
Dr. Ann Marie Navar noted that she received consulting fees from Merck during the study period. She also disclosed consulting fees from other pharmaceutical companies that manufacture various lipid-lowering therapies. The study was funded by Merck Sharp & Dohme, a subsidiary of Merck.
The senior investigators, including Drs. Brown, Goldstein, Hobbs, and Cohen, maintain significant academic appointments at UT Southwestern, holding various distinguished chairs that underscore their roles in the ongoing history of cholesterol research.
Conclusion: A New Era of Heart Health
The potential for enlicitide to enter the market represents a convergence of decades of scientific discovery. By targeting the PCSK9 pathway—the same biological mechanism identified by the UTSW team decades ago—but delivering it in a user-friendly oral format, enlicitide addresses the limitations that have long plagued lipid management.
For the millions of Americans navigating the risk of heart disease, the future of treatment is looking increasingly manageable. If the upcoming clinical outcomes trials confirm that these cholesterol reductions lead to fewer cardiovascular events, enlicitide could move from an experimental pill to a standard-of-care medication, fundamentally reshaping how physicians approach the prevention of the world’s leading cause of death. As the medical community awaits the final outcomes data, the message remains clear: we are closer than ever to a future where heart attacks and strokes are not just managed, but effectively preempted.