For most of human history, the sense of smell has been shrouded in a biological mystery. While our eyes rely on a mere three types of photoreceptors to paint the world in color, and our ears translate mechanical vibrations into the symphony of sound, the nose functions as a chemical detective, navigating a vast, invisible landscape of molecules. It is a sense that warns us of fire, flavors our food, and acts as a direct, visceral conduit to our deepest memories and emotions. Yet, despite its profound influence on human experience, the underlying biological "map" of olfaction has remained frustratingly elusive—until now.
In a landmark study published in the journal Cell on April 28, researchers at Harvard Medical School have finally decoded the spatial organization of the olfactory system. By analyzing millions of neurons in mice, the team has revealed that the nose is not a chaotic, randomly organized sensory organ, but rather a highly structured landscape. This discovery marks a fundamental shift in our understanding of how the brain perceives the chemical world and offers a long-awaited roadmap for addressing olfactory dysfunction.
The Great Sensory Mystery
"Olfaction is super-mysterious," says Sandeep (Robert) Datta, professor of neurobiology at the Blavatnik Institute at Harvard Medical School and the senior author of the study. For decades, neuroscientists have struggled to reconcile the complexity of smell with the lack of apparent organization in the olfactory epithelium—the tissue inside the nose responsible for detecting scents.
While vision, hearing, and touch were mapped with relative ease, smell remained the "odd one out." The reason for this gap in knowledge lies in the sheer, staggering scale of the system. Humans and mice alike possess a vast repertoire of smell receptors. A mouse, for instance, utilizes over a thousand distinct receptor types to identify an near-infinite variety of odorants. Each neuron expresses only one type of receptor, and there are approximately 20 million olfactory neurons in the mouse nose.
For years, the scientific consensus was that these neurons were distributed in a broad, semi-random fashion. Because researchers could not identify a clear structural pattern, they concluded that the system lacked the geometric precision found in other sensory organs. This assumption, however, proved to be incorrect.
A Chronology of Discovery: From Randomness to Order
The quest to map the nose began in earnest in 1991, when researchers first identified the genes that encode smell receptors. Over the ensuing decades, the scientific community attempted to determine whether these receptors were clustered by function or location. Early experiments, constrained by the technology of the time, suggested that receptors appeared in only a few wide, undefined zones. This led to the pervasive, yet flawed, notion that the olfactory map was essentially stochastic.
As high-throughput genetic sequencing and spatial transcriptomics emerged, the Datta lab saw an opportunity to revisit this foundational question. The team embarked on a massive undertaking: analyzing approximately 5.5 million neurons across more than 300 mice.
"This is now arguably the most sequenced neural tissue ever, but we needed that scale of data in order to understand the system," Datta explains. By combining single-cell sequencing—which identifies the specific receptor expressed by each neuron—with spatial transcriptomics—which tracks the exact physical location of those neurons—the researchers were finally able to see the forest for the trees.
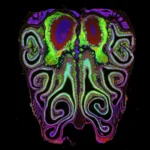
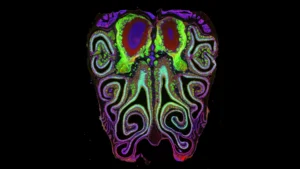
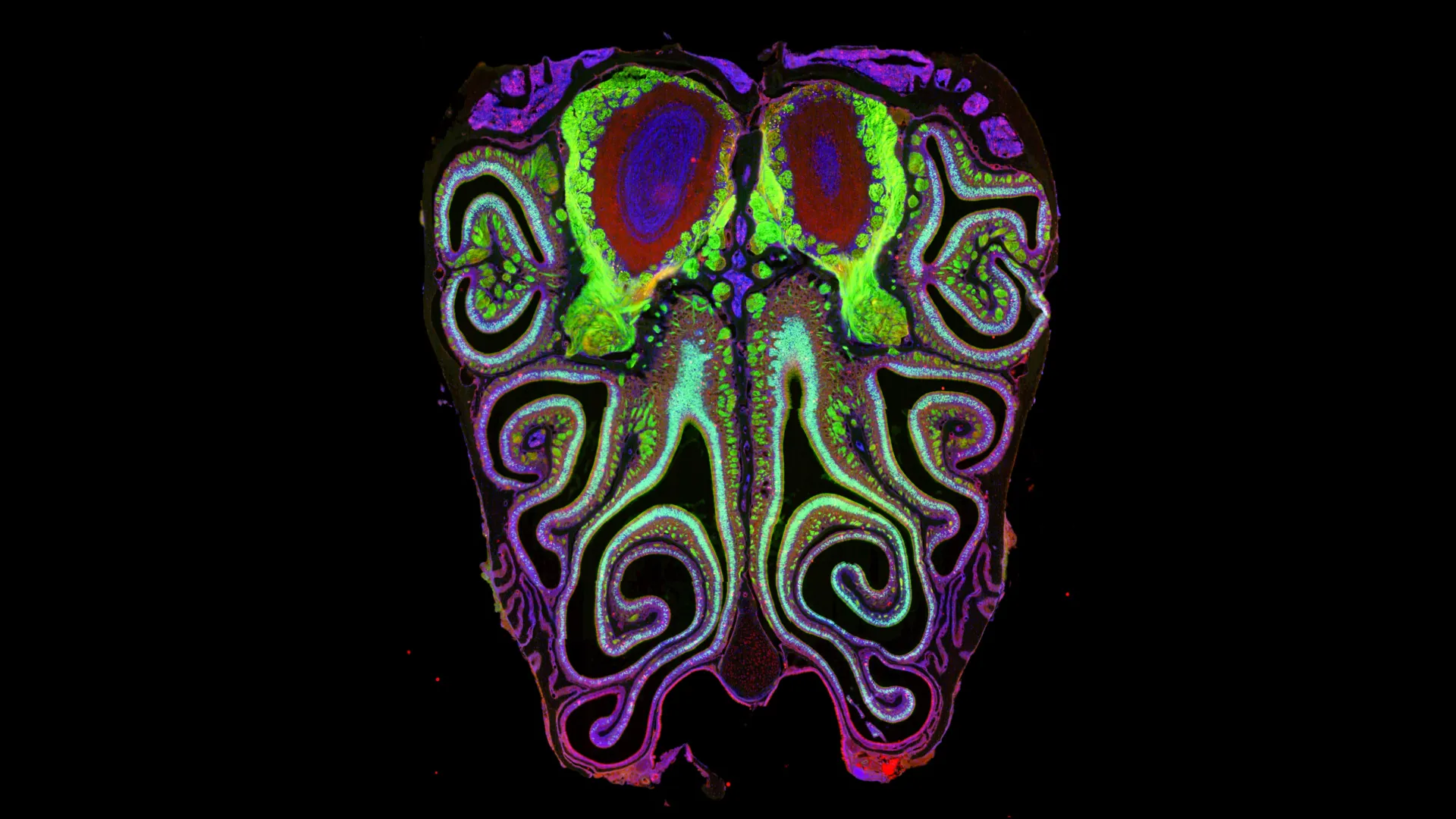
The results were transformative. The data revealed a highly consistent, repeating pattern of horizontal bands or "stripes" running from the top to the bottom of the nose. These stripes are not haphazard; they are meticulously organized by receptor type. This structure is not a quirk of a single specimen but a universal architecture that remains nearly identical across different animals.
Decoding the Mechanism: The Role of Retinoic Acid
Once the team confirmed the existence of this rigid architectural map, the next logical question was: How does it form? The development of such a complex, precise, and consistent map requires a sophisticated biological instruction manual.
The research team identified a chemical gradient—specifically, a gradient of retinoic acid—as the primary architect of this system. Retinoic acid, a molecule known for its role in gene regulation during embryonic development, appears to act as a molecular GPS for developing neurons. By varying the levels of retinoic acid, the body dictates which smell receptors are activated based on a neuron’s physical position in the nasal cavity.
To prove this, the team performed a series of experiments in which they artificially altered the concentrations of retinoic acid. The results were dramatic: the entire receptor map shifted upward or downward in response to the changes. This confirmed that the "stripes" are not merely a static feature but a dynamically regulated biological system.
"We show that development can achieve this feat of organizing a thousand different smell receptors into an incredibly precise map that’s consistent across animals," Datta remarked. In the same issue of Cell, a parallel study led by the laboratory of Catherine Dulac, the Xander University Professor at Harvard, provided independent, corroborating evidence for these findings, further solidifying the discovery.
Implications for Clinical Medicine and Human Health
The implications of this study extend far beyond the realm of basic developmental biology. The loss of smell—often associated with viral infections, neurodegenerative diseases, or physical trauma—is a condition that currently lacks effective medical interventions. For the millions of people who suffer from anosmia (the loss of smell) or hyposmia (the reduced ability to smell), the impact is not merely limited to the loss of culinary enjoyment.
Olfaction is deeply intertwined with safety, as it allows us to detect smoke, gas leaks, and spoiled food. Furthermore, its profound connection to the limbic system—the part of the brain that governs memory and emotion—means that a loss of smell can have severe psychological repercussions, contributing to depression and social withdrawal.
"We cannot fix smell without understanding how it works on a basic level," Datta emphasizes. By establishing the "ground truth" of the olfactory map, researchers can now begin to approach the problem of restoration with greater precision. If scientists can understand how the nose is wired, they may eventually be able to use stem cell therapies or advanced brain-computer interfaces to rewire or regenerate damaged olfactory circuits.
The research team is already looking toward the next phase: determining whether this horizontal striping pattern is conserved in humans and investigating the specific functional roles of these individual stripes.
Conclusion: A New Frontier in Neuroscience
The Harvard team’s findings effectively dismantle the long-standing belief that the sense of smell is a disorganized sensory system. By proving that the nose follows a rigorous, genetically controlled blueprint, the study has provided the field of neurobiology with a new framework for exploration.
"Smell has a really profound and pervasive effect on human health, so restoring it is not just for pleasure and safety but also for psychological well-being," Datta concludes. "Without understanding this map, we’re doomed to fail in developing new treatments."
As science continues to peel back the layers of the olfactory system, the "mysterious" sense is finally beginning to make sense. For those living in a world deprived of scent, this map offers more than just academic clarity; it offers the flickering, distant hope of a future where the sense of smell can be understood, managed, and perhaps one day, restored.
Authorship, Funding, and Disclosures
This study was a collaborative effort involving researchers David Brann, Tatsuya Tsukahara, Cyrus Tau, Dennis Kalloor, Rylin Lubash, Lakshanyaa Kannan, Nell Klimpert, Mihaly Kollo, Martin Escamilla-Del-Arenal, Bogdan Bintu, Andreas Schaefer, Alexander Fleischmann, and Thomas Bozza.
Financial support for this research was provided by the National Institutes of Health (grants R01DC021669, R01DC021422, R01DC021965, and F31DC019017), the Yang Tan Collective at Harvard, and a National Science Foundation Graduate Research Fellowship.