BARCELONA, Spain — For millions of patients living with heart failure (HF), the hospital ward is an all-too-familiar, often demoralizing, landscape. The cycle of fluid overload, breathlessness, and emergency admission for intravenous (IV) diuretic therapy has long been the gold standard for stabilizing these patients. However, a landmark trial presented at the European Society of Cardiology’s Heart Failure 2026 meeting suggests that the future of heart failure care may lie not in a hospital bed, but in the comfort of a patient’s own home.
The SUBCUT II HF trial has demonstrated that patients suffering from worsening heart failure can be safely and effectively transitioned to ambulatory care using a wearable minipump that delivers subcutaneous furosemide. By allowing patients to manage their congestion outside the clinical environment, the study points toward a transformative shift in how clinicians approach one of the most common and taxing conditions in cardiovascular medicine.
The Core Innovation: Moving Beyond the IV Drip
At the heart of this shift is an infusor device designed for simplicity and autonomy. Attaching to the abdominal skin with standard medical adhesive, the device allows patients to self-administer furosemide—a potent diuretic—at the push of a button. The medication itself, Lasix Onyu (SQ Innovation), was formulated specifically for subcutaneous delivery and received FDA approval in October 2025.
Unlike traditional hospital-based IV treatment, which requires round-the-clock nursing supervision, this "patch-pump" technology empowers patients to manage their diuretic needs once or twice daily. The device builds upon the promising, yet limited, findings of the earlier SUBCUT HF I pilot study, scaling the concept to a more robust, multi-center trial that challenges the necessity of prolonged hospital stays for congestion management.
Chronology of a Paradigm Shift
The journey to the results presented in Barcelona was methodical and patient-centered. Investigators sought to answer a fundamental question: Could the safety and efficacy of hospital-based IV therapy be replicated in a home setting without compromising patient outcomes or increasing mortality?
The trial, conducted across 22 National Health Service (NHS) hospitals in the United Kingdom, enrolled 172 patients with a mean age of 71. The cohort represented a high-acuity population, with 92% suffering from peripheral edema and 29% experiencing pleural effusion. The average NT-proBNP level—a key biomarker for heart failure severity—was 3,365 pg/mL, underscoring the significant burden of congestion among participants.
The Study Protocol
Participants were randomized into two distinct cohorts:
- Usual Care (n=80): Patients remained in the hospital setting, receiving standard-of-care IV furosemide until medical staff determined their fluid levels had stabilized.
- Early Discharge (n=92): Patients were trained on the use of the subcutaneous minipump before being discharged early. Once home, they received 80 mg of furosemide via the pump once or twice daily, supplemented by a study visit every three days to monitor their progress.
Both groups underwent a comprehensive clinical review within two weeks of discharge to ensure no resurgence of symptoms occurred.
Data and Clinical Outcomes
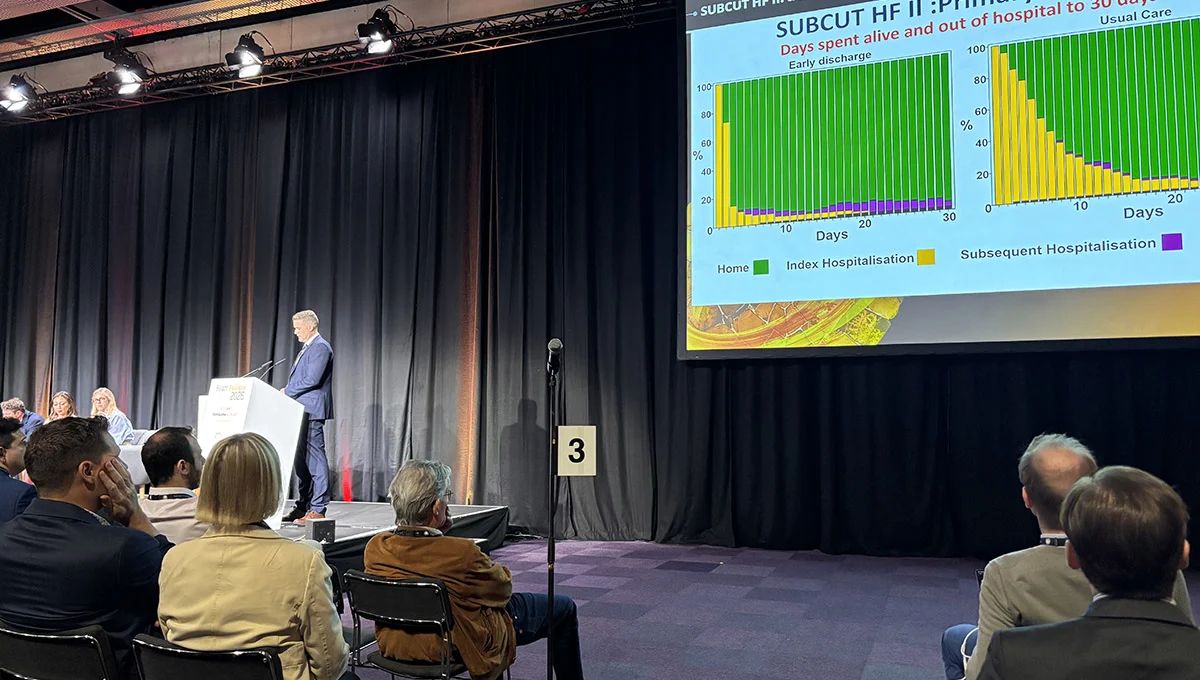
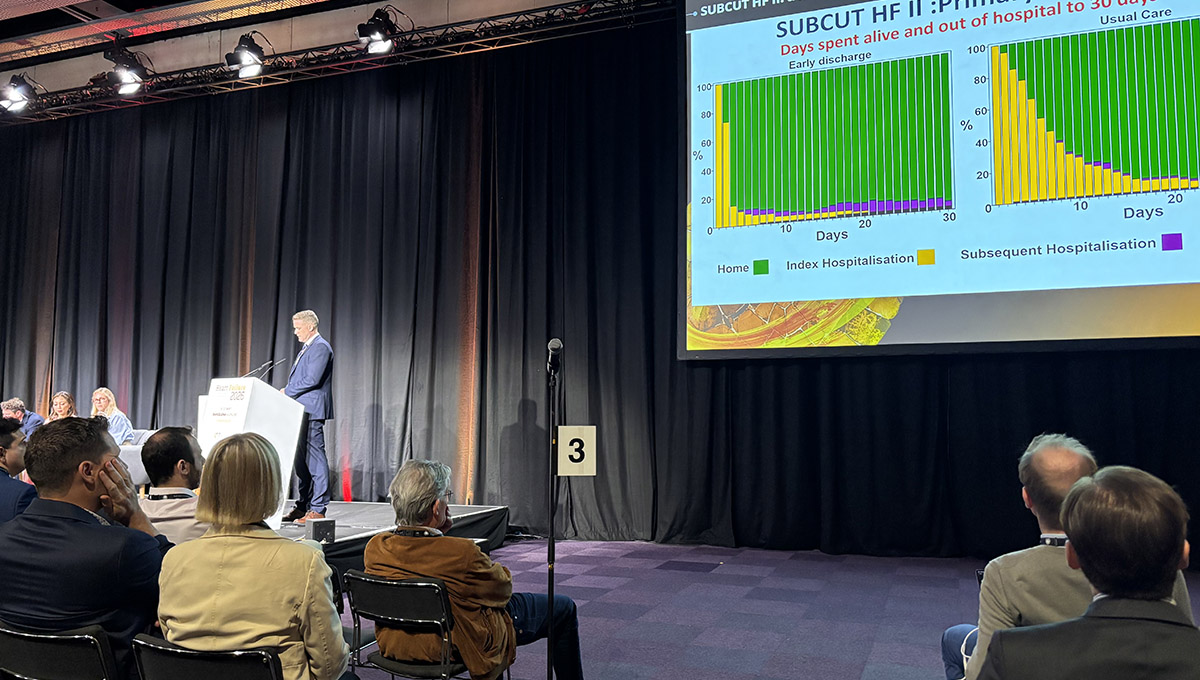
The primary outcome of the trial—measured as the number of days spent alive and out of the hospital within 30 days of randomization—was a clear victory for the early-discharge strategy. Patients in the intervention group spent an average of 4.0 days more out of the hospital compared to their counterparts receiving standard care (95% CI 2.1-5.8; P < 0.001).
The benefits were equally apparent at the 60-day mark. Patients in the early-discharge group enjoyed a significantly shorter index hospital stay, averaging 5.6 days versus 11.0 days for the standard-care group. Furthermore, by day 60, the early-discharge group had spent an average of 53.9 days alive and out of the hospital, compared to 49.7 days for those who remained in the traditional inpatient ward.
Importantly, these gains were achieved without sacrificing patient well-being. Quality-of-life metrics, measured via the Kansas City Cardiomyopathy Questionnaire (KCCQ)-12, showed no significant difference between the two groups. Additionally, there were no significant discrepancies in the rates of cardiovascular death or subsequent heart failure hospitalizations, suggesting that the home-based approach was as robust as its inpatient equivalent.
Perspectives from the Clinical Community
The trial’s investigators were initially cautious. Ross Campbell, MBChB, PhD, and Mark Petrie, MBChB, both of the University of Glasgow, admitted that the transition from continuous hospital supervision to intermittent monitoring was met with some anxiety.
"We were worried," Dr. Petrie noted during his presentation. "We thought people might go home and be rehospitalized a lot, die, or have complications because we only saw them every three days in the intervention arm, as opposed to the continuous care provided in a hospital setting."
Those fears proved unfounded. The safety profile of the pump was excellent; there were no device-related serious adverse events throughout the study. Of the 1,054 doses administered, more than 90% were handled directly by patients or their caregivers, proving that the technology is intuitive and accessible to the demographic most affected by heart failure.
However, the medical community remains balanced in its appraisal. Dr. Jan Biegus of Wroclaw Medical University, while praising the study as a "positive" and "difficult to conduct" piece of research, offered a necessary note of skepticism. He pointed out that the trial was open-label, which introduces an unavoidable risk of bias.
Furthermore, Dr. Biegus noted the "built-in advantage" of the intervention group. Because they were discharged early by design, they essentially started the clock with a head start on "days out of hospital." Despite this, he acknowledged that the results remain compelling. "More people prefer being at home rather than in the hospital, so this is the way forward," he concluded.
Implications for Future Heart Failure Care
The successful implementation of the SUBCUT II HF protocol suggests that the healthcare system is on the precipice of a new era. The implications of this trial extend far beyond the immediate goal of discharging patients a few days earlier.
A Spectrum of Potential Uses
Dr. Petrie emphasized that the utility of subcutaneous furosemide extends well beyond the early-discharge model. "We see lots of roles for it," he noted.
- Preventative Management: For patients in the community who are showing early signs of fluid accumulation, the pump could be deployed to nip congestion in the bud, preventing an emergency room visit or a full-scale hospitalization before it ever becomes necessary.
- Palliative Care: For patients in end-of-life care, the pump provides a discreet, non-invasive way to manage the discomfort of fluid overload, allowing them to remain in a home setting rather than being subjected to the intensity of an acute care ward.
The Need for Strategic Patient Selection
Despite the excitement, investigators stressed that this therapy is not a "one-size-fits-all" solution. The success of the trial depended heavily on patient selection. To be candidates for this program, patients must demonstrate a baseline level of physical capability, possess sufficient cognitive clarity to operate the device, and—crucially—have a support system at home, whether it be a partner, family member, or visiting caregiver.
Looking Ahead
The landscape for at-home heart failure management is rapidly evolving. With the FDA approval of other subcutaneous furosemide delivery systems, such as Furoscix, the availability of these tools is increasing. The next major milestone will be a cluster-randomized trial of Lasix Onyu in the United States, which aims to further validate these findings across a larger, more diverse patient population.
As healthcare costs continue to climb and the global population ages, the ability to transition high-acuity care into the home environment is no longer just a luxury—it is a necessity. The SUBCUT II HF trial provides a roadmap for how we might treat patients with more dignity, greater comfort, and ultimately, better clinical efficiency. For the patient with heart failure, the "hospital of the future" may soon be their own living room.