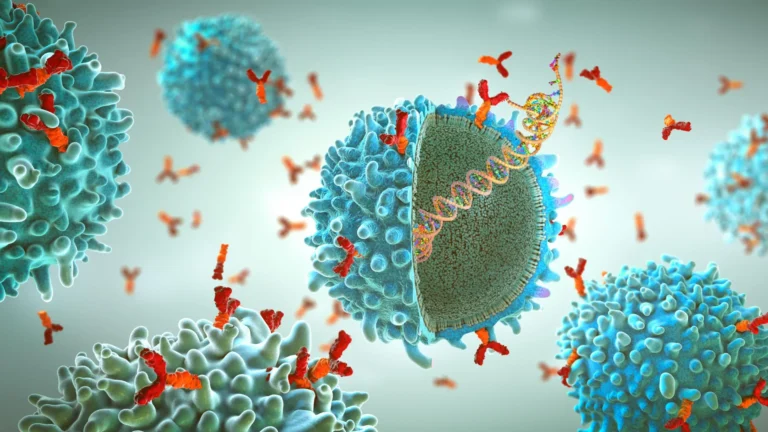
In a landmark achievement for immunology and vaccine safety, an international team of researchers—led by Flinders University and including scientists from Greifswald University and McMaster University—has successfully identified the biological trigger behind a rare but dangerous blood clotting condition associated with certain adenovirus-based vaccines.
The discovery, recently published in the New England Journal of Medicine, marks the final chapter in a years-long global scientific investigation. By pinpointing exactly how the immune system misidentifies a viral protein as a threat to the body’s own blood-clotting mechanism, researchers have paved the way for the next generation of safer, genetically refined vaccines.
The Core Discovery: Molecular Mimicry Unmasked
The condition in question, known as Vaccine-Induced Immune Thrombocytopenia and Thrombosis (VITT), emerged as a clinical enigma during the global rollout of COVID-19 vaccines in 2021. Characterized by low platelet counts and the formation of dangerous blood clots, VITT presented a significant safety hurdle for health authorities.
For years, the scientific community struggled to understand why a tiny fraction of individuals experienced this adverse event. The latest study provides the definitive answer: Molecular Mimicry.
Researchers discovered that the human immune system can, in rare instances, mistake a specific protein found in the adenovirus—the vector used to deliver vaccine instructions—for Platelet Factor 4 (PF4), a protein naturally occurring in human blood. When the immune system detects the adenovirus, it produces antibodies to neutralize the perceived threat. However, due to the structural similarities between the viral protein and PF4, these antibodies cross-react, binding to and activating the body’s own platelets. This "immune system mix-up" triggers an unregulated clotting cascade, leading to the clinical manifestations of VITT.
Dr. Jing Jing Wang, a lead researcher at Flinders University, described the breakthrough as the "missing link" in understanding how a healthy immune response can, in extraordinary circumstances, go awry. "By using powerful mass spectrometry sequencing, we were able to visualize the exact point where this molecular mimicry occurs," Dr. Wang stated. "This provides a clear explanation for how a normal, protective immune response turns into a pathogenic, autoimmune event."
A Chronology of Investigation: From Discovery to Resolution
The road to this discovery was not linear; it was a multi-year effort that required international cooperation and a series of rigorous scientific milestones.
2021: The Emergence of VITT
As COVID-19 vaccination programs expanded, rare reports of atypical blood clots surfaced in patients who had received adenovirus-based vaccines, most notably the Oxford-AstraZeneca (Vaxzevria) vaccine. The medical community quickly identified that these clots were linked to autoantibodies targeting PF4, but the reason why the immune system targeted this specific blood protein remained unknown.
2022: The Genetic Connection
A crucial step forward occurred in 2022 when Dr. Wang and Professor Tom Gordon, Head of Immunology at SA Pathology, decoded the structural profile of the PF4 antibody. Their research revealed a critical genetic susceptibility factor: a specific antibody gene variant known as IGLV3.2102. This finding suggested that while the vaccine was the catalyst, an individual’s genetic makeup played a significant role in their predisposition to the reaction.
2023: The Natural Infection Parallel
The scope of the research expanded in 2023 when Professor Ted Warkentin from McMaster University reported that VITT-like conditions were not exclusive to vaccines. His team observed near-identical reactions in patients suffering from natural adenovirus infections (the common cold). This was a pivotal realization: it proved that the issue was inherent to the adenovirus itself, rather than a side effect of any specific vaccine manufacturing process or additive.
2024: Closing the Trilogy
The 2024 follow-up study, which serves as the culmination of this international collaboration, provided the final piece of the puzzle. By proving that antibodies from both vaccine-related and natural infection-related cases were indistinguishable, the team confirmed that the adenovirus pVII protein was the primary culprit.
The Mechanics of the "Mix-Up"
To understand the severity of this discovery, one must look at the function of Platelet Factor 4. Under normal conditions, PF4 is a protein released by platelets that helps facilitate blood coagulation and wound healing. It is a vital component of the body’s self-repair system.
In the case of VITT, the immune system’s defensive antibodies—designed to protect the body from the viral vector—accidentally lock onto the PF4. This binding creates a "super-antigen" complex that sends a false signal to the platelets, telling them to aggregate and form clots even when there is no injury to repair. Because the body is simultaneously consuming its own platelets to form these unnecessary clots, the patient’s overall platelet count drops (thrombocytopenia), leading to a paradox of excessive clotting and a simultaneous inability to stop bleeding.
The identification of the pVII protein as the specific trigger allows developers to move from observation to mitigation. By modifying the genetic sequence of the adenovirus vector to exclude or alter the pVII protein, vaccine manufacturers can effectively "remove the disguise," ensuring that the immune system focuses entirely on the intended viral target without cross-reacting with PF4.
Official Responses and Scientific Impact
The academic community has hailed the findings as a landmark in medical science. Professor James McCluskey, a renowned immunologist from the University of Melbourne and the Peter Doherty Institute, praised the work as a "brilliant piece of molecular sleuthing."
"This research represents the culmination of a massive, multi-year effort to unravel the genetic and structural basis of pathogenic autoimmunity," Professor McCluskey said. "It is rare that we can trace a clinical phenomenon all the way down to the atomic level of protein structure, but that is exactly what this team has achieved."
Professor Tom Gordon, who has been at the forefront of this research since the onset of the pandemic, reflected on the importance of the global partnership. "It has been a fascinating journey with an outstanding international team of collaborators. We have effectively solved the mystery of this new group of blood clotting disorders. More importantly, we have translated our discoveries into a tangible path for future vaccine safety."
The Flinders University team emphasized that the findings do not diminish the value of adenovirus-based vaccines, which have saved millions of lives during the pandemic and continue to be a pillar of public health in many developing nations. Instead, this discovery provides the technical blueprints to optimize these platforms for future use.
Implications for Global Public Health
The potential for this discovery to improve global vaccine technology cannot be overstated. Adenovirus-based vectors are highly efficient at inducing immune responses and are relatively inexpensive to produce, making them an ideal candidate for combating infectious diseases in resource-limited settings.
1. Safer Vaccine Engineering
By editing the pVII protein, developers can create vaccines that retain their high efficacy while eliminating the potential for the VITT reaction. This is a significant step toward restoring public confidence in viral-vector technology.
2. Broadening Therapeutic Applications
The discovery of how molecular mimicry can trigger autoimmunity has implications beyond vaccines. Understanding this pathway may provide insights into other autoimmune disorders where the body’s immune response is triggered by environmental or viral factors.
3. Policy and Regulatory Guidance
Health regulatory bodies, including the Therapeutic Goods Administration (TGA) in Australia and the FDA in the United States, now have a clear, evidence-based mechanism to evaluate the safety of future vector-based vaccines. This transparency allows for more nuanced risk-benefit analyses and the potential for targeted screening programs for individuals who may carry the IGLV3.2102 genetic marker.
As the world looks toward future pandemic preparedness, the "trilogy" of research led by Flinders University, Greifswald, and McMaster provides a masterclass in how international scientific collaboration can address urgent global health crises. By turning a complex medical mystery into a solvable molecular problem, these researchers have ensured that the next generation of vaccines will be safer, more targeted, and better equipped to protect the global population.
The story of VITT, which began as a source of confusion and alarm, has ultimately become a testament to the power of persistent, collaborative scientific inquiry. As vaccine developers begin the work of refining viral vectors, the legacy of this research will be felt in the safety and efficacy of the medicines of tomorrow.