In the high-stakes theater of human immunology, few actors are as formidable or as precise as the cytotoxic T lymphocyte (CTL). Often described as the "serial killers" of the immune system, these specialized white blood cells are tasked with a mission of surgical lethality: patrolling the body to identify, infiltrate, and destroy cells compromised by viral infection or malignant transformation.
For decades, the mechanism by which these cells execute their target has been understood in broad strokes, but the fine architectural details of this interaction—the "immune synapse"—have remained elusive. Now, a groundbreaking study from the University of Geneva (UNIGE) and the Lausanne University Hospital (CHUV) has pierced this veil. By leveraging a revolutionary imaging technique known as cryo-expansion microscopy (cryo-ExM), researchers have successfully mapped the internal machinery of T cells in three dimensions, capturing them in their near-native state. The findings, published in the journal Cell Reports, offer a transformative look at how the immune system orchestrates its most lethal maneuvers, potentially providing the blueprint for the next generation of immuno-oncology therapies.
The Immune Synapse: A Microscopic Battlefield
At the core of the immune response lies the immune synapse, a highly organized, transient interface formed when a CTL encounters a target cell. This is not a simple collision; it is a sophisticated molecular handshake. Once the CTL identifies a foreign antigen presented on the surface of a target cell, it establishes a tight, polarized contact zone.
Within this confined space, the T cell secretes "cytotoxic granules"—packets containing lethal proteins like perforin and granzymes. Perforin creates pores in the target cell membrane, while granzymes enter to trigger programmed cell death, or apoptosis. This process must be executed with extreme fidelity; if the toxic molecules were released haphazardly, they would cause catastrophic collateral damage to healthy neighboring tissues. While the functional outcome of this synapse is well-documented, the structural "scaffolding" required to maintain such precision has long frustrated microscopists.
A History of Imaging Hurdles
The challenge in visualizing the immune synapse has historically been a clash between resolution and preservation. To see the nanometer-scale architecture of a living cell, researchers have traditionally relied on electron microscopy, which requires harsh chemical fixation and dehydration—processes that inevitably warp and distort the delicate, fluid-like structures of the cell membrane.
Alternatively, conventional light microscopy provides better context but lacks the resolution to distinguish the minute structural shifts occurring at the moment of cell-to-cell contact. For years, the scientific community has been forced into a "trilemma": researchers had to choose between high resolution, a wide field of view, or the preservation of the cell’s natural, "living" state. This inability to see the "real" architecture of the synapse meant that many of the nuanced structural triggers of T-cell activation remained purely theoretical.
Cryo-Expansion Microscopy: A Technological Leap
To bypass these limitations, the collaborative team from UNIGE and CHUV-UNIL, supported by the ISREC Foundation TANDEM program, turned to cryo-expansion microscopy (cryo-ExM). This hybrid approach represents a significant evolution in cellular imaging.
How the Process Works
The technique is a two-fold innovation. First, the samples are subjected to "vitrification"—an instantaneous freezing process at extremely high speeds. By cooling the cells rapidly, the water within them turns into a glass-like solid rather than expanding into ice crystals, which would otherwise puncture and destroy cellular membranes.
Second, the vitrified samples are infused with an absorbent, expandable hydrogel. As the gel expands, it physically stretches the cellular architecture outward without distorting the spatial relationships of the internal organelles. "This technique involves instantaneously freezing cells… where water solidifies without forming crystals and thus faithfully preserves biological structures," explains Virginie Hamel, Senior Lecturer in the Department of Molecular and Cellular Biology at the Faculty of Science of UNIGE. "The samples are then physically expanded… making it possible to observe their internal organization with great precision while maintaining their near-native architecture."
Unveiling the "Dome": New Structural Discoveries
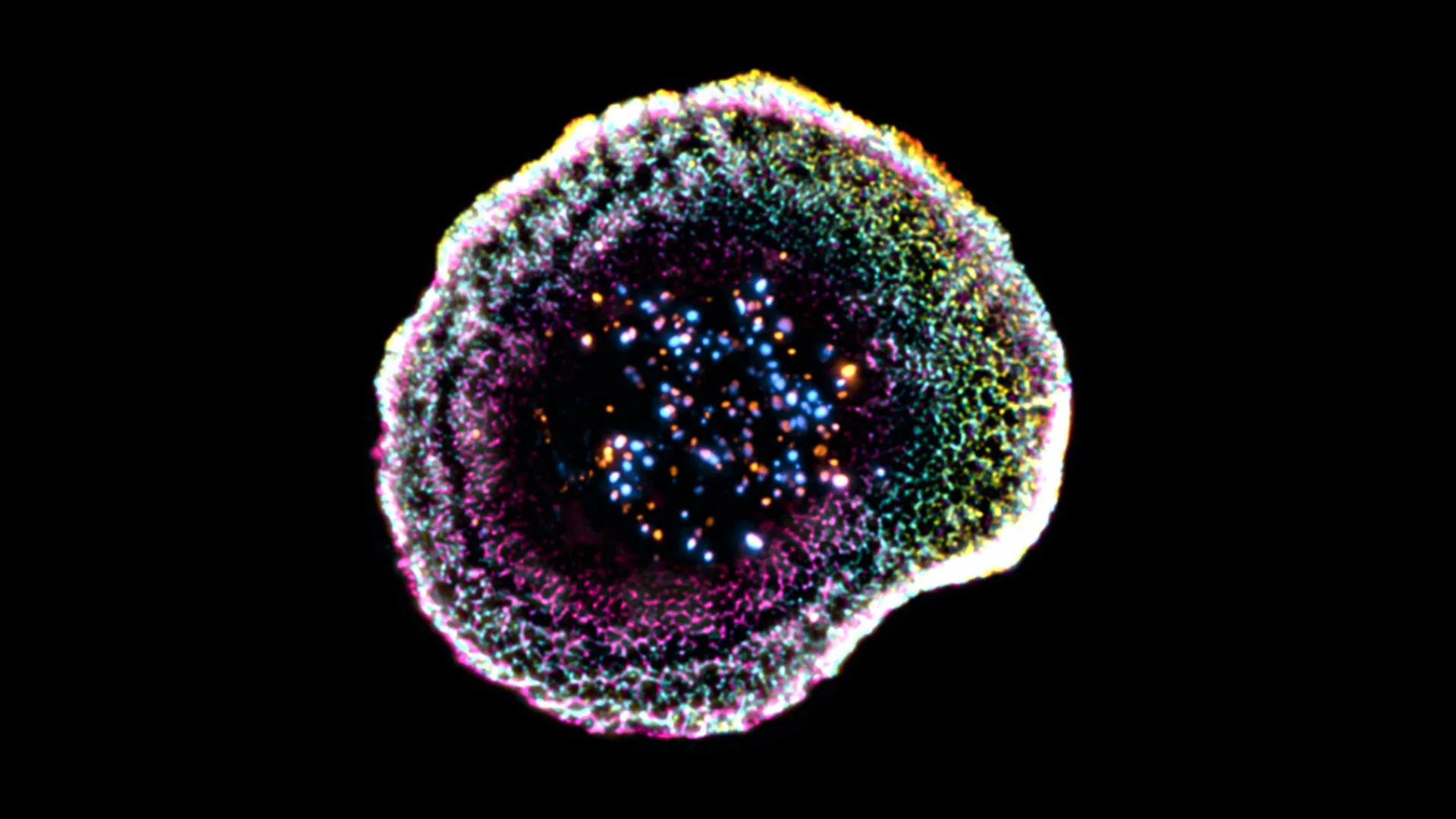
The application of cryo-ExM yielded immediate and startling results. The researchers were able to visualize the contact zone between the T cell and its target with unprecedented clarity, revealing that the immune synapse is not merely a flat interface.
The Dome Architecture
"Our work reveals that at the point of contact between the immune cell and its target, the membrane forms a kind of dome," notes Florent Lemaître, a postdoctoral researcher at UNIGE and the study’s lead author. This "dome" structure is not accidental; it appears to be physically linked to the internal cytoskeletal organization of the T cell and the adhesion molecules that anchor it to the target. This structural curvature suggests that the T cell actively shapes its membrane to optimize the delivery of its lethal payload, ensuring a hermetic seal that prevents the escape of toxic molecules into the surrounding environment.
The Anatomy of Cytotoxic Granules
Beyond the membrane, the team also gained a clearer view of the cytotoxic granules themselves. Previously, these were often viewed as uniform, spherical vesicles. The 3D imaging revealed that these granules possess internal heterogeneity. Some contain multiple "cores" where active molecules are concentrated, while others appear more uniform. This discovery raises new questions: does the internal structure of these granules correlate with their potency or the speed at which they trigger apoptosis? The ability to see these sub-vesicular details provides a new metric for assessing the "readiness" of a T cell to engage in combat.
Clinical Implications: From Lab Cells to Human Tumors
Perhaps the most significant aspect of this study is its transition from controlled laboratory cultures to the messy, complex environment of actual human tumor tissues. In clinical settings, T cells are often exhausted or blocked by the tumor microenvironment.
"We extended this approach to human tumor tissues, making it possible to directly observe T lymphocytes infiltrating tumors and their cytotoxic machinery at the nanometer scale," says Benita Wolf, Chief Resident and Associate Researcher in the Department of Clinical Oncology at CHUV, who co-led the study.
By applying cryo-ExM to real-world tumor samples, the researchers have created a diagnostic framework that allows oncologists to look at how a patient’s own immune system is interacting with their cancer. If a patient is failing to respond to immunotherapy, it may now be possible to visualize whether their T cells are failing to form the proper "dome" at the synapse, or if their cytotoxic granules are structurally deficient.
The Future of Immuno-Oncology
The implications for the future of medicine are profound. Immuno-oncology has revolutionized the treatment of cancer, but it remains a game of probabilities; treatments like CAR-T cell therapy work brilliantly for some and not at all for others.
By providing a three-dimensional, high-resolution map of the immune synapse in action, the UNIGE/CHUV team has provided the industry with a new "standard of proof." Researchers can now test how various drugs or genetic modifications influence the physical assembly of the synapse. For instance, if a new therapy aims to boost T-cell efficacy, scientists can use cryo-ExM to see if that therapy actually leads to more robust dome formation or more efficiently organized granules.
Furthermore, this study bridges the gap between basic cell biology and clinical pathology. It transforms the T cell from an abstract entity into a visible, mechanical machine. As scientists continue to refine this imaging technique, the ability to observe the "killer" cells of the immune system in their natural, lethal state will likely accelerate the development of personalized cancer treatments that are more precise, more effective, and less toxic to the patient.
In the quest to master the body’s defenses, we are finally moving beyond watching the war from a distance. With cryo-ExM, we are now looking at the microscopic front lines, watching the immune system do what it does best: defend, strike, and conquer—one nanometer at a time.