By [Your Name/Journalistic Desk]
May 11, 2026
In the high-stakes world of federal health policy, few projections carry as much weight—or cause as much internal anxiety—as the annual spending estimates for Medicare. When the Centers for Medicare & Medicaid Services (CMS) and its actuaries turn their attention to breakthrough therapies, the numbers they produce serve as the bedrock for federal budget planning, drug pricing negotiations, and private-sector investment strategies.
Two years ago, the medical and financial communities braced for an "Alzheimer’s shock." Following the accelerated approval of Leqembi (lecanemab), a monoclonal antibody developed by Eisai and Biogen, analysts predicted a massive fiscal burden on the Medicare program. Projections released in 2024 suggested that Medicare would spend approximately $3.5 billion on the drug in 2025 alone. Today, however, as fresh data rolls in, it has become clear that these predictions were profoundly wide of the mark. The anticipated fiscal tsunami has, thus far, been a ripple.
The Evolution of a Forecast: A Chronology of Expectation vs. Reality
To understand why the spending gap exists, one must look at the timeline of the Alzheimer’s drug rollout.
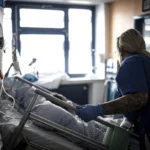
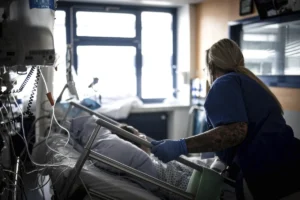
- January 2023: The FDA grants accelerated approval to Leqembi, signaling a new era in neurodegenerative disease treatment. The initial enthusiasm is tempered by questions regarding accessibility, the complexity of infusion administration, and the rigorous diagnostic requirements (PET scans or cerebrospinal fluid tests) necessary to confirm amyloid plaque presence.
- April 2024: As the drug gains broader clinical acceptance, Medicare actuaries release updated forecasts. The $3.5 billion figure for 2025 becomes the standard benchmark for fiscal planning, reflecting a belief that patient uptake would accelerate rapidly as infusion centers increased capacity.
- Late 2024 – Early 2025: Bottlenecks emerge. While the science of the drug holds, the "real-world" logistics of identifying, diagnosing, and treating patients prove significantly more cumbersome than federal models accounted for.
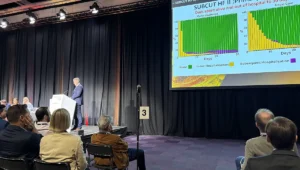
- May 2026: New data indicates that actual Medicare spending on Leqembi and its successor in the class, Kisunla (donanemab), is substantially lower than the 2024 estimates. The "spending gap" becomes the primary topic of conversation among health policy wonks in Washington.
Supporting Data: Where the Models Diverged
The discrepancy between the $3.5 billion projection and current spending levels stems from a combination of clinical, logistical, and behavioral factors that federal models historically struggle to capture.
1. The Diagnostic Chokepoint
The most significant barrier to rapid adoption has not been the cost of the drug itself, but the requirements for its administration. To qualify for Leqembi or Kisunla, patients must undergo expensive, time-consuming confirmation of amyloid pathology. Many rural hospitals and smaller clinics lack the specialized imaging infrastructure (amyloid PET scans) or the capacity to perform lumbar punctures. This has created a geographic disparity in access, effectively capping the number of eligible patients who can actually receive the therapy.
2. The Infusion Burden
Unlike traditional pharmaceuticals that can be picked up at a pharmacy, these Alzheimer’s treatments require bi-weekly or monthly intravenous infusions. This creates a significant "time cost" for patients and caregivers. Medicare’s models often assume a linear adoption curve based on the number of diagnosed patients; they fail to account for the reality that many elderly patients, particularly those in the early stages of cognitive decline, may find the logistical burden of recurring clinic visits prohibitive.

3. Clinician Hesitancy and Patient Selection
Neurologists and geriatricians have been more cautious than market analysts predicted. The safety profile of these drugs—which includes risks of ARIA (amyloid-related imaging abnormalities)—has led to a rigorous selection process. Physicians are not prescribing these drugs to every patient with mild cognitive impairment; they are engaging in extensive risk-benefit discussions that reduce the total volume of patients initiating treatment.
Official Responses and Industry Perspectives
The pharmaceutical industry, specifically Eisai and its partners, has maintained that their focus remains on patient access rather than short-term revenue spikes. In recent investor calls, leadership has highlighted the "long-term buildup" of the Alzheimer’s market.
"We are building a new standard of care from the ground up," an industry spokesperson noted. "It is not a matter of whether the market will grow, but rather how we solve the infrastructure challenges that keep patients from receiving the life-changing therapies they deserve."
Conversely, CMS has been relatively tight-lipped regarding the specific mechanics of their forecasting errors, though internal memos suggest a shift toward more "dynamic" modeling. The agency is now looking at how to better integrate real-world evidence—such as infusion center wait times and regional imaging availability—into their actuarial processes.
"Predicting the future of health care spending is notoriously difficult," said one former health policy advisor. "When you introduce a completely new class of drugs that requires a total overhaul of the patient journey, your baseline assumptions are almost certainly going to be wrong."
Implications for the Health Care Landscape
The failure of the $3.5 billion projection has several critical implications for the broader health care system.
The Budgetary "Safety Net"
For the federal government, the lower-than-expected spending is, on the surface, a fiscal win. It alleviates immediate pressure on the Medicare Part B trust fund. However, it also suggests that the government is under-prepared for what might happen if diagnostic bottlenecks are cleared. If the Biden or future administrations move to subsidize the diagnostic infrastructure, we could see a sudden, rapid surge in utilization that the budget has not accounted for.

Drug Pricing Negotiations
The Inflation Reduction Act (IRA) allows Medicare to negotiate prices for certain high-spend drugs. By falling short of the spending thresholds, these Alzheimer’s drugs may stay off the "negotiation radar" for longer than originally anticipated. This provides the manufacturers with a window of relative price stability, but it also creates a political narrative that the high list prices are effectively "self-policing" because of low utilization.
The Innovation Cycle
For biotech and pharma investors, the lesson is clear: scientific success does not equate to commercial success. The "commercialization valley of death"—the gap between FDA approval and widespread adoption—is wider for neurodegenerative diseases than it is for oncology or rare diseases. Future investment in Alzheimer’s research will likely be tempered by the realization that infrastructure, not just efficacy, drives the bottom line.
Conclusion: The Road Ahead
As we look toward the latter half of 2026, the Alzheimer’s market remains in a state of adjustment. The "spending gap" is a reminder that the U.S. health care system is a complex, often glacial, machine. While the technology for treating Alzheimer’s exists, the human and logistical systems required to deliver it are still in their infancy.
The error in the $3.5 billion forecast should not be viewed as a failure of math, but as a lesson in the complexity of medical innovation. Medicare will continue to refine its models, and manufacturers will continue to push for broader access. But for now, the data tells a story of a revolution that is happening, just much more slowly than anyone in Washington—or Wall Street—dared to predict.
Do you have insights on how Medicare’s forecasting errors are impacting your practice or region? Reach out to our investigative team to share your story.