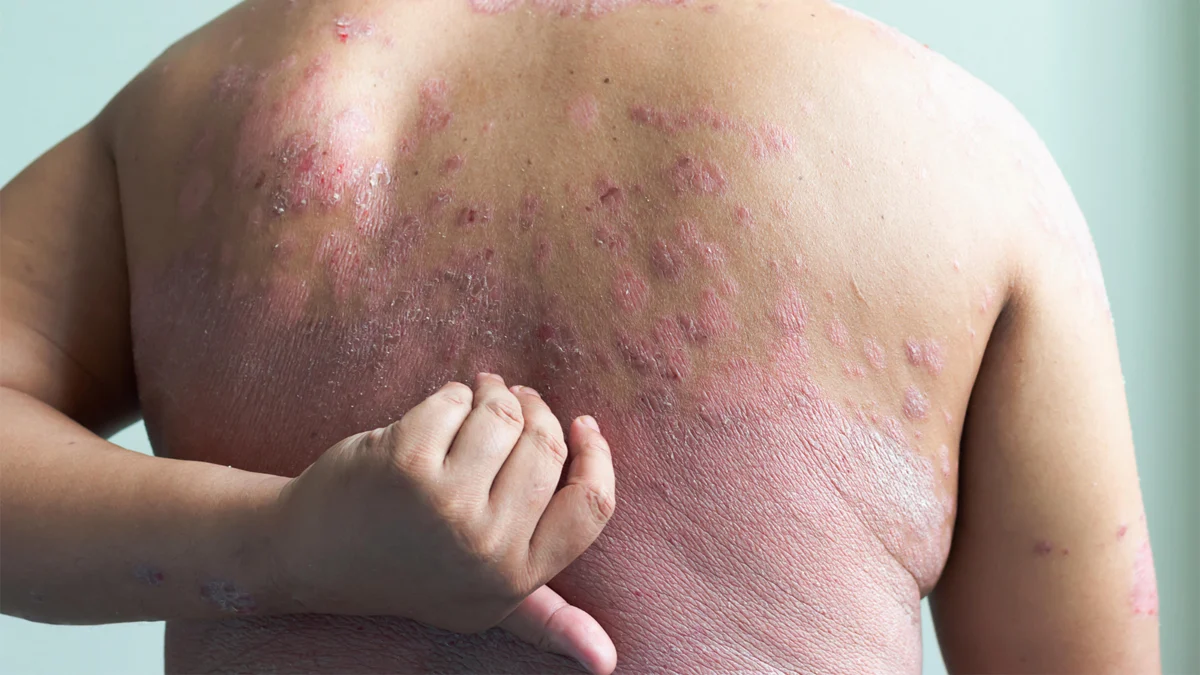
In a landmark shift for dermatological care, a new phase IIIb study has unveiled a promising new paradigm for treating plaque psoriasis. The TOGETHER-PsO trial, recently published in JAMA Dermatology and presented at the Society for Investigative Dermatology annual meeting in Chicago, suggests that combining the psoriasis biologic ixekizumab (Taltz) with the dual GLP-1/GIP receptor agonist tirzepatide (Zepbound) significantly outperforms traditional monotherapy in patients grappling with both moderate-to-severe plaque psoriasis and obesity.
The findings indicate that the future of psoriasis management may lie in addressing the systemic, inflammatory, and metabolic roots of the disease rather than treating the skin in isolation.
The Main Facts: A Dual-Action Success
The TOGETHER-PsO trial aimed to tackle one of the most stubborn clinical challenges: managing psoriasis in patients with a high body mass index (BMI). Because obesity itself is a pro-inflammatory state that can hinder the efficacy of biologic treatments, researchers sought to determine if systemic metabolic correction could improve skin outcomes.
At the 36-week mark, the data provided a definitive answer. Researchers, led by Dr. Mark Lebwohl of the Icahn School of Medicine at Mount Sinai, reported that 27.1% of patients receiving the combination therapy achieved the primary endpoint—a composite of complete skin clearance (PASI 100) and at least a 10% reduction in total body weight. In stark contrast, only 5.8% of patients receiving ixekizumab alone reached this threshold, a difference that proved statistically significant (P<0.001).
Beyond the composite endpoint, the combination therapy showed superiority in skin clearance alone. Patients on the dual regimen achieved a 40.6% rate of PASI 100, compared to 29% in the monotherapy group (P=0.04). These results suggest that by reducing systemic inflammation through metabolic regulation, clinicians can "unlock" the full potential of traditional biologics.
Chronology: The Evolution of the TOGETHER-PsO Study
The trial was a rigorous 52-week investigation conducted across 72 sites in the United States. It recruited 274 adults, all of whom presented with moderate-to-severe plaque psoriasis, an average disease duration of nearly 15 years, and a high baseline BMI (mean 39.2).
- Baseline (Week 0): Patients were randomized 1:1 to receive either ixekizumab plus tirzepatide or ixekizumab alone.
- The Midpoint (Week 36): The primary efficacy data were measured, showing a massive divergence in both weight loss and skin-clearing efficacy between the two groups.
- The Full Duration (Week 52): The trial concluded with comprehensive safety and metabolic reporting, confirming that the benefits extended beyond the skin into blood pressure, triglyceride levels, and patient-reported quality of life.
This trial follows on the heels of the TOGETHER-PsA study, which investigated similar interventions for psoriatic arthritis. Together, these trials provide a growing body of evidence suggesting that incretin-based therapies—originally designed for diabetes and weight management—are becoming essential tools in the rheumatology and dermatology toolkit.
Supporting Data: The Metabolic-Dermatological Link
The depth of the trial data underscores the systemic nature of psoriatic disease. The researchers monitored not only skin scores but also critical cardiovascular and metabolic biomarkers.
Key Efficacy Metrics
| Metric | Ixekizumab + Tirzepatide | Ixekizumab Alone | P-Value |
|---|---|---|---|
| PASI 100 + 10% Weight Loss | 27.1% | 5.8% | <0.001 |
| PASI 100 | 40.6% | 29.0% | 0.04 |
| PASI 75 + 10% Weight Loss | 69.2% | 9.1% | <0.001 |
| 10%+ Weight Reduction | 69.0% | 9.1% | <0.001 |
The trial also demonstrated significant improvements in metabolic health, including reductions in blood pressure and triglycerides, which are notoriously high in patients with psoriasis. Importantly, these improvements were achieved with a safety profile that did not present any novel or unexpected risks. While gastrointestinal events (common with GLP-1/GIP agonists) were more frequent in the combination group, they were generally mild to moderate, and the rates of serious adverse events were comparable to the control group.
Official Responses and Clinical Perspectives
The medical community has reacted with significant enthusiasm to the study, though some have offered measured caution regarding the study’s design.
A Call to Action for Dermatologists
In an accompanying editorial, Michael Garshick, MD, MS, a cardiologist at NYU School of Medicine, and Joel Gelfand, MD, MSCE, a dermatologist at the University of Pennsylvania, lauded the study as a "call to action." They argued that the current standard of care—which often treats psoriasis as a strictly dermatological condition—is outdated.
"Cardiovascular disease in patients with psoriasis starts early with endothelial damage… potentiated by proinflammatory cytokines, dyslipidemia, insulin resistance, and overactivated platelets," the authors wrote. They noted that despite the clear link between psoriasis and cardiometabolic morbidity, these patients remain consistently undertreated for their cardiovascular risk factors.
Addressing Limitations
The editorialists were careful to note that the study’s open-label design and lack of a placebo-controlled arm for the tirzepatide component mean that further research is needed. They proposed an intriguing, radical shift in the care algorithm: "For some patients, particularly those with obesity and mild to moderate psoriasis, it is possible that their psoriasis may remit with incretin agonists alone." This would establish a new paradigm where metabolic therapy serves as the first-line treatment, with traditional skin-targeted biologics reserved for patients who do not achieve full remission.
Implications: A New Era in Psoriatic Disease Management
The implications of the TOGETHER-PsO trial are far-reaching, potentially reshaping how insurance companies, clinicians, and regulatory bodies view the treatment of chronic inflammatory conditions.
1. From "Skin-Deep" to "Systemic"
Psoriasis has long been categorized as a systemic immune-mediated disease, but treatment has remained largely focused on the skin. By proving that a dual-action therapy can simultaneously improve skin, joint, and metabolic health, the medical community is moving toward a more holistic model of care.
2. Expanding the Indications of Incretins
The rapid expansion of GLP-1 and dual-receptor agonist indications—ranging from metabolic dysfunction-associated steatohepatitis (MASH) to sleep apnea and cardiovascular disease—suggests these drugs may be the "statins of the 21st century." Their role in dermatology is likely to grow as the National Psoriasis Foundation and other bodies begin to formalize guidelines for their use.
3. Addressing the "Psoriatic Comorbidity Gap"
Patients with psoriasis are at higher risk for heart disease, diabetes, and obesity-related complications. By utilizing a drug that directly addresses the patient’s BMI while reducing the inflammatory cytokines that fuel plaque growth, clinicians can effectively treat the "whole patient." This not only improves quality of life scores but may, in the long run, reduce the heavy burden of mortality and morbidity associated with chronic psoriatic disease.
4. Future Research Needs
While the results are compelling, the scientific community is already looking toward the next phase. Large-scale, randomized, placebo-controlled trials will be necessary to establish standardized dosing, long-term safety profiles, and the ideal patient profile for combination therapy. Researchers are also eager to see if these findings translate across different biologic classes, or if the synergy is unique to the interaction between IL-17 inhibitors like ixekizumab and the GLP-1/GIP pathway.
Conclusion
The TOGETHER-PsO trial marks a watershed moment in dermatology. By integrating metabolic medicine with targeted immunotherapy, researchers have opened a door to a more effective, comprehensive, and patient-centered approach to psoriasis. As the lines between metabolic health and immunology continue to blur, the medical community stands on the precipice of a new era—one where clearing the skin is merely the beginning of restoring the patient’s overall health. The evidence is clear: the future of psoriasis treatment is not just skin deep; it is systemic, metabolic, and transformative.