By Medical News Staff | May 7, 2026
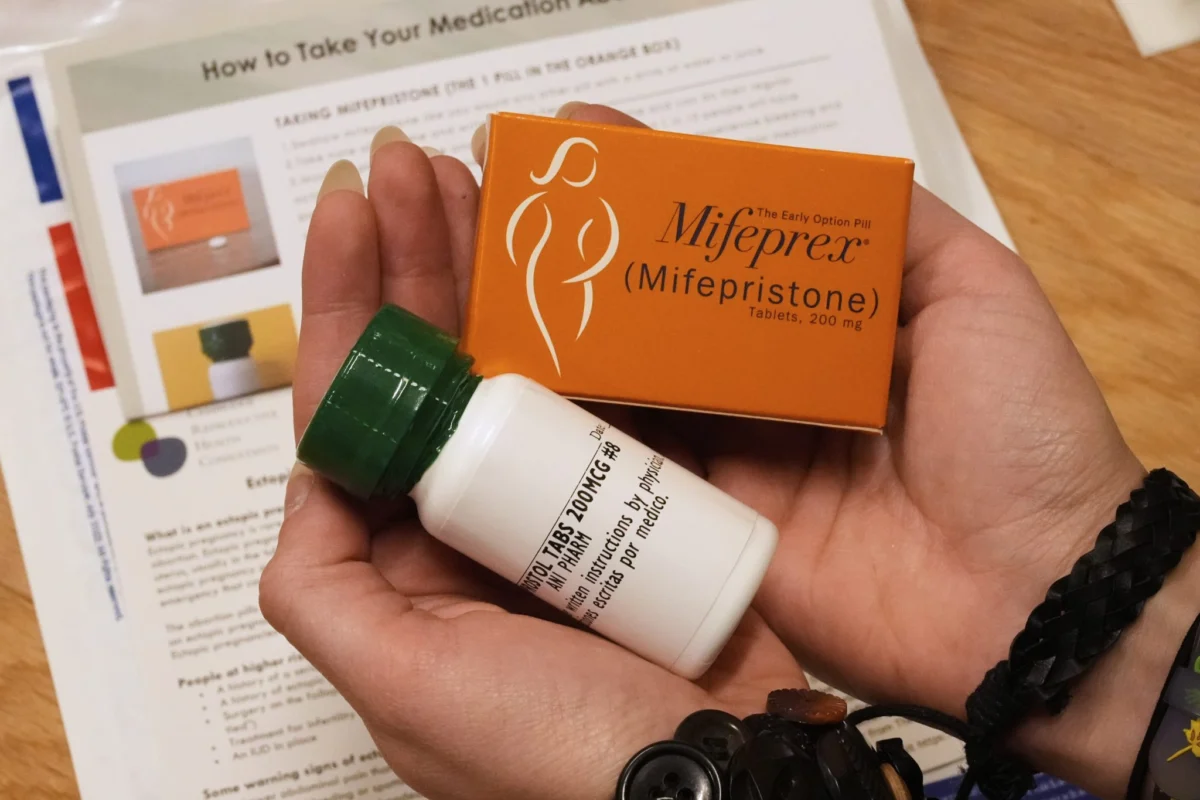
The landscape of reproductive healthcare in the United States faces a pivotal transformation following a high-stakes legal intervention by the 5th U.S. Circuit Court of Appeals. On May 1, 2026, a three-judge panel issued a ruling that immediately mandates in-person dispensing for mifepristone—the most widely utilized medication for abortion—effectively halting the mail-order delivery model that has served as a cornerstone of telehealth-based reproductive care since the height of the COVID-19 pandemic.
The ruling has sent shockwaves through the medical community, leaving patients, clinicians, and telehealth providers in a state of operational uncertainty. While the Supreme Court has issued a temporary administrative stay to preserve the status quo through mid-May, the long-term future of medication abortion access remains caught in a volatile tug-of-war between federal regulatory authority and state-level legislative mandates.
Chronology: A Legal Escalation
The current crisis did not emerge in a vacuum; it is the culmination of years of litigation aimed at dismantling the FDA’s long-standing approval process for mifepristone.
- 2021–2023: Following the COVID-19 public health emergency, the FDA permanently lifted in-person dispensing requirements for mifepristone, citing clinical data confirming the safety of remote consultations and mail delivery.
- April 2026: A federal district court declined to restrict access to the drug, noting that the FDA was currently conducting a safety review under the directive of the Trump administration.
- May 1, 2026: The 5th U.S. Circuit Court of Appeals reversed the district court’s decision. The panel ruled that federal agency actions permitting mail-order mifepristone effectively undermined state-level abortion bans, specifically citing Louisiana’s restrictive laws.
- May 2, 2026: Danco Laboratories, the manufacturer of the brand-name medication Mifeprex, filed an emergency appeal with the U.S. Supreme Court, requesting an immediate pause on the appellate court’s ruling.
- May 4, 2026: Supreme Court Justice Samuel Alito issued a one-week administrative stay, temporarily blocking the 5th Circuit’s requirement for in-person visits. This stay is set to expire on May 11, 2026, creating a narrow window for the Court to decide whether to hear the merits of the case.
Supporting Data: The Shift to Telehealth
To understand the gravity of this ruling, one must examine how abortion care has shifted in the wake of the 2022 Dobbs v. Jackson Women’s Health Organization decision. According to data from the Guttmacher Institute, medication abortion now accounts for 63% of all abortions in the United States, a significant increase from 53% in 2020.
The rise of telehealth has been the primary driver of this shift. For many patients, particularly those in "abortion deserts" or states with restrictive laws, the ability to consult with a provider via video conference and receive medication by mail has been the only viable path to care.
The Guttmacher Institute’s research suggests that the removal of mail-delivery options would disproportionately affect vulnerable populations. Clinical experts emphasize that the necessity of traveling to a physical clinic—often requiring time off work, childcare, and travel expenses—creates an insurmountable barrier for rural patients, individuals with low incomes, and those living with disabilities.
The Constitutional Conflict: Federal vs. State Authority
At the heart of the 5th Circuit’s decision is a fundamental question of jurisdiction. The court argued that the FDA’s regulatory flexibility regarding mifepristone acts as a federal override of state-level bans. The ruling states that federal agency actions "cancel" the efficacy of state-level restrictions, suggesting that the FDA’s authority to approve and regulate drugs cannot supersede a state’s right to enforce its own medical abortion bans.
This logic, however, is heavily contested by legal scholars and the Biden-era FDA officials. They maintain that federal drug approval is a national standard and that the FDA’s decision to allow mail-order delivery was based on comprehensive clinical data, not political preference. Furthermore, the court noted that the FDA’s current, ongoing safety review—initiated under the Trump administration—has yet to reach a conclusion, with officials admitting that they are still in the data-gathering phase.
Official Responses and Stakeholder Concerns
The legal community is deeply divided on the implications of the 5th Circuit ruling.
Danco Laboratories’ Stance:
In its emergency filing to the Supreme Court, Danco Laboratories warned that the appellate court’s decision "injects immediate confusion and upheaval into highly time-sensitive medical decisions." The company underscored the clinical reality that medical abortion is a time-sensitive procedure, and any disruption to the delivery of care can result in patients missing the window of efficacy for medication-based interventions.
Advocacy and Clinical Perspectives:
Julia Kaye, an attorney with the ACLU, expressed severe concerns regarding the impact on equity. "Restricting telemedicine harms rural communities, people with low incomes, people with disabilities, survivors of intimate partner violence, and communities of color most severely," Kaye told NPR.
Medical associations have similarly sounded the alarm, arguing that the court’s decision ignores the scientific consensus on the safety of mifepristone. They emphasize that the medication has a long track record of safety, and that the physical location of the patient at the time of ingestion does not correlate with clinical outcomes.
Implications for Telehealth Clinicians
For clinicians currently operating in the reproductive health space, the current climate is one of extreme caution. The legal status of mifepristone is in flux, and providers are facing a "patchwork" regulatory environment.
Operational Challenges
- Prescription Complications: Existing prescriptions and pending mail-order shipments are currently in legal limbo. Pharmacies are struggling to determine whether they can legally fulfill orders without violating the 5th Circuit’s potential mandate.
- Scheduling Instability: Telehealth platforms are currently re-evaluating their intake processes. Many are informing patients that while services continue under the Supreme Court’s stay, the situation could change overnight on May 11.
- Liability Concerns: Clinicians are advised to consult with their state medical boards and legal counsel. The conflict between federal regulatory guidance and state-level court mandates creates a high risk of litigation for providers who continue to offer remote dispensing.
Recommendations for Providers
Experts suggest that clinicians remain in close contact with legal counsel and professional liability carriers. Key steps include:
- Monitoring State Board Guidance: As states react to the federal court rulings, local boards may issue specific instructions that override general federal guidelines.
- Transparent Patient Communication: Providers must ensure that patients are fully informed of the current legal risks and the potential for service disruptions, particularly for those who have not yet received their medication.
- Documenting Care: In this litigious environment, meticulous documentation of the clinical rationale for medication abortion and the adherence to current, active federal regulations is essential.
Conclusion: A Decisive Moment for Healthcare Access
The battle over mifepristone is more than a dispute over drug distribution; it is a battle over the future of decentralized, technology-driven healthcare in America. If the Supreme Court ultimately sides with the 5th Circuit, the impact will be a structural dismantling of the telehealth abortion model, forcing millions back into a clinic-based system that is increasingly inaccessible.
As the May 11 deadline approaches, the nation watches to see if the Supreme Court will maintain the administrative stay or allow the 5th Circuit’s restrictive ruling to take full effect. For the millions of patients who rely on the privacy and accessibility of telehealth, the coming weeks will determine whether they retain control over their reproductive health, or whether the geography of their residence will once again dictate their fundamental medical rights.