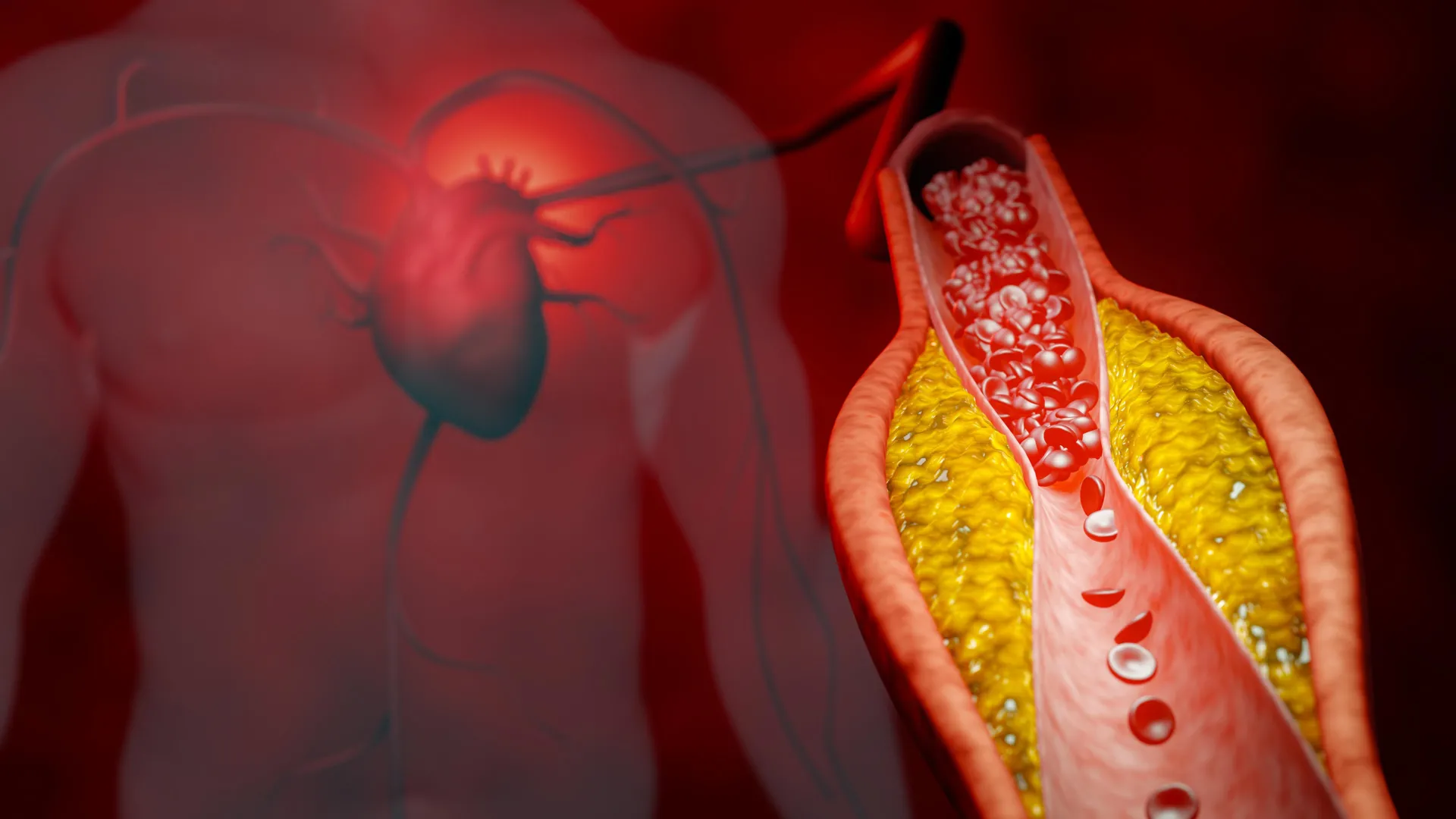
High cholesterol—specifically elevated levels of low-density lipoprotein (LDL-C)—remains one of the most pervasive health crises of the modern era. Often termed "hypercholesterolemia," this condition acts as a silent architect of cardiovascular decay, slowly depositing fatty plaques into the walls of the arteries, a process known as atherosclerosis. This narrowing of the vessels sets the stage for heart attacks, strokes, and peripheral artery disease, claiming millions of lives annually.
While the medical community has long relied on statins—a class of drugs that inhibit cholesterol production in the liver—these treatments are not without their limitations. Many patients suffer from debilitating side effects, such as muscle pain (myopathy), while others find their cholesterol levels stubbornly resistant to conventional pharmacology. Now, a collaborative team of international researchers from the University of Barcelona and the University of Oregon has unveiled a pioneering approach that targets the genetic root of the problem: the PCSK9 protein.
The Science of the "Bad" Cholesterol Regulator
At the heart of the research is PCSK9 (proprotein convertase subtilisin/kexin type 9), a protein that serves as a metabolic gatekeeper. Under normal physiological conditions, the liver uses LDL receptors to "sweep" LDL cholesterol out of the bloodstream. However, PCSK9 has a destructive affinity for these receptors; it binds to them and triggers their degradation. When PCSK9 levels are high, the liver loses its ability to clear cholesterol, leaving the "bad" fat to circulate freely and accumulate within arterial walls.
The research team, led by Professors Carles J. Ciudad and Verònica Noé of the University of Barcelona’s Faculty of Pharmacy and Food Sciences, in collaboration with Dr. Nathalie Pamir of the University of Oregon, has developed a novel intervention: polypurine hairpins (PPRHs). These specialized DNA-based molecules function as genetic "silencers," blocking the production of PCSK9 before it can ever be synthesized.
Chronology of Discovery: From Bench to Breakthrough
The development of the PPRH therapy represents the culmination of years of molecular investigation. The journey began with the identification of specific genomic targets within the PCSK9 gene structure. By analyzing the genetic sequence, researchers sought a way to interrupt transcription—the process by which DNA instructions are copied into RNA—without utilizing the complex and sometimes immunogenic delivery mechanisms required by traditional gene therapies.
- Target Identification: Researchers focused on exons 9 and 12 of the PCSK9 gene. These segments were selected for their accessibility to synthetic oligonucleotide interventions.
- Molecular Engineering: The team synthesized two distinct polypurine hairpins, dubbed HpE9 and HpE12. These hairpins were engineered to bind precisely to their target DNA sequences through Watson-Crick base pairing.
- In Vitro Validation: The team tested these molecules on HepG2 liver cells. The initial results were striking, showing significant inhibition of both PCSK9 RNA and protein production.
- In Vivo Testing: Recognizing the potential for clinical application, the team transitioned to transgenic mouse models expressing the human PCSK9 gene. This stage confirmed that the therapy was not only effective in a petri dish but also viable in a living biological system.
- Peer Review and Publication: Following rigorous testing and replication, the findings were officially documented and published in the journal Biochemical Pharmacology, marking the formal entry of PPRHs into the cardiovascular research lexicon.
Supporting Data: Quantitative Efficacy
The power of the HpE12 hairpin, in particular, has caught the attention of the scientific community due to its unprecedented efficiency. According to the study, the molecular mechanism works by interfering with the activity of RNA polymerase or essential transcription factors. When these factors are blocked, the PCSK9 gene remains "switched off."
The statistical evidence provided by the researchers is compelling:
- Cellular Impact: In HepG2 liver cell cultures, HpE12 demonstrated an 87% reduction in PCSK9 protein levels and a 74% reduction in PCSK9 RNA levels.
- Systemic Impact (Animal Models): In the transgenic mouse model, a single administration of the treatment resulted in a 50% decrease in plasma PCSK9 levels.
- Cholesterol Reduction: Perhaps most significantly, the therapy led to a 47% reduction in total cholesterol levels in the mice within just 72 hours of administration.
These figures suggest that PPRHs could be substantially more potent than many current pharmacological interventions, which often require daily dosing and carry the risk of cumulative systemic toxicity.
Official Perspectives: The Experts Speak
The lead researchers have been vocal about the significance of these findings, particularly in how they distinguish this therapy from existing cholesterol-lowering drugs.
"Specifically, one of the arms of each chain of the HpE9 and HpE12 polypurines binds specifically to polypyrimidine sequences of exons 9 and 12 of PCSK9, respectively, via Watson-Crick bonds," explains Professor Carles J. Ciudad. By precisely targeting these sequences, the hairpins effectively stall the transcriptional machinery.
Professor Verònica Noé emphasizes the practical benefits for patients: "The results show that both HpE9 and HpE12 are highly effective. Beyond the raw data, the stability and low cost of synthesis for these oligonucleotides represent a major leap forward in therapeutic design."
The research was made possible through the support of the Spanish Ministry of Science, Innovation and Universities (MICINN) and the National Institutes of Health (NIH) in the United States. This international cooperation underscores the global urgency of finding safer, more effective alternatives to current statin-based regimens.
Implications for Future Cardiovascular Care
The implications of this study are profound, particularly for patients who have found current treatments insufficient or intolerable. While the market currently features monoclonal antibodies like evolocumab and alirocumab, as well as the siRNA-based therapy Inclisiran, these treatments face hurdles related to cost, delivery, and the necessity of repeat administration.
Advantages of the PPRH Approach
- Low Cost of Synthesis: Unlike monoclonal antibodies, which require complex biological manufacturing processes, PPRHs are simpler and cheaper to produce, potentially making them more accessible to global healthcare systems.
- Safety and Stability: PPRHs have demonstrated a lack of immunogenicity—the body does not recognize them as foreign invaders to the same degree as other gene-silencing therapies, reducing the risk of allergic or immune-mediated side effects.
- Avoidance of Statins: By bypassing the metabolic pathways targeted by statins, PPRHs avoid the common and often painful side effect of myopathy. This is a critical factor for patient adherence, as many individuals discontinue statin therapy due to muscle-related discomfort.
- Targeted Precision: The ability of HpE12 to achieve such high knockdown rates with a single injection suggests that the frequency of dosing could be dramatically lower than current daily medication requirements.
Challenges Ahead
Despite the optimism surrounding these results, the research team is careful to note that clinical translation is a rigorous process. Before human trials can begin, further studies must address long-term safety, potential off-target effects, and the optimization of delivery systems to ensure the DNA hairpins reach the liver cells efficiently in human patients.
Furthermore, the team must determine the duration of the effect. While the 47% reduction in cholesterol observed in the mouse model is highly promising, long-term monitoring will be required to ensure that these levels remain stable over months or years of treatment.
Conclusion: A New Frontier in Preventive Medicine
The development of polypurine hairpins represents a shift toward "genetic pharmacology"—a future where medicine is not merely masking symptoms, but actively correcting the molecular imbalances that lead to disease. By neutralizing the PCSK9 protein at the transcriptional level, Professors Ciudad, Noé, and Pamir have opened a door to a potential "cure" for high-risk cholesterol profiles that are currently difficult to manage.
As the study moves toward its next phases, the medical community remains hopeful. If these results are replicated in human clinical trials, we may be looking at a future where cardiovascular disease is significantly mitigated, not just by managing cholesterol levels, but by fundamentally reprogramming the liver’s ability to maintain its own health. In the ongoing war against heart disease, the HpE12 hairpin may prove to be one of the most precise and powerful weapons ever forged in a laboratory.