By Ben Fidler | Published May 14, 2026
The quest to provide a definitive treatment for Duchenne muscular dystrophy (DMD)—a devastating, progressive, and fatal neuromuscular condition—has reached a critical, if precarious, juncture. Regenxbio, a biotechnology company heavily invested in the promise of gene therapy, is currently maneuvering through a landscape defined by shifting regulatory sands, safety concerns, and the long, complicated shadow cast by its predecessor in the space, Sarepta Therapeutics’ Elevidys.
As of May 2026, Regenxbio’s experimental gene therapy, RGX-202, has hit key clinical milestones. However, the path to market remains obscured by the emergence of two serious adverse events and a volatile leadership transition within the U.S. Food and Drug Administration (FDA). Despite these headwinds, CEO Curran Simpson remains steadfast, betting that the agency’s internal mandate for “rare disease flexibility” will ultimately pave the way for a 2027 approval.
Main Facts: The Promise and The Peril
At the heart of the debate is the mechanism of RGX-202. Like the currently available Elevidys, RGX-202 is designed to deliver a functional gene to the body that enables the production of microdystrophin—a crucial "shock absorber" protein that DMD patients lack, leading to the gradual necrosis of muscle fibers.
Where RGX-202 aims to differentiate itself is in its delivery and design. The therapy utilizes a distinct viral vector platform and produces a larger, potentially more stable version of the microdystrophin protein. Furthermore, the company has implemented a specialized regimen of immune-suppressing drugs intended to mitigate the inflammatory responses that have plagued other gene therapy programs.
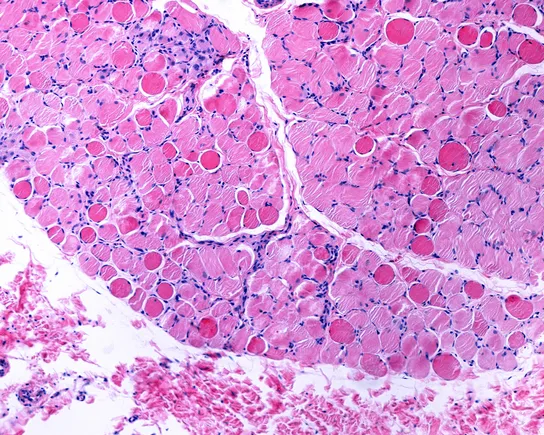
However, clinical development in Duchenne is notoriously difficult. The field is still reeling from the controversial approval of Elevidys, which, despite achieving accelerated clearance, faced significant backlash due to underwhelming efficacy data and subsequent reports of potentially fatal liver-related side effects. The cooling of the market for Elevidys serves as both a cautionary tale and a competitive vacuum that Regenxbio hopes to fill with a more robust clinical profile.
A Chronology of Clinical Development
The trajectory of RGX-202 has been defined by a series of strategic alignments with the FDA, though the regulatory environment has shifted drastically since those initial discussions took place.
- Early Development: Regenxbio initiated its trials with the premise that its unique viral vector and protein design would yield superior efficacy compared to existing standards.
- Alignment with the FDA: The company successfully negotiated an abbreviated approval path, opting to expand a single-arm study rather than engaging in the arduous process of a large-scale placebo-controlled trial—a hurdle that has caused other major players, such as Pfizer, to falter.
- The 10% Benchmark: The primary goal of the expanded study was to demonstrate the ability of RGX-202 to boost microdystrophin levels to at least 10% of normal expression.
- 2026 Data Readout: In May 2026, the company confirmed it had cleared its primary threshold. Microdystrophin expression averaged approximately 71% across the cohort. Notably, in boys over the age of 8—a demographic where the disease trajectory typically takes a sharp turn for the worse—the expression levels remained significant at 42%.
- Current Standing: The company is now in a "wait and see" posture, delaying its formal Biologics License Application (BLA) to gauge the direction of the FDA under its current leadership turnover.
Supporting Data: The Case for Efficacy
Regenxbio’s recent data release serves as the strongest argument for the therapy’s potential. By demonstrating a mean microdystrophin expression of 71%, the company has provided the FDA with a surrogate marker that, in theory, should correlate with long-term clinical benefit.
Most importantly, the company claims to have established a statistical link between this protein expression and a slowing of the disease’s natural trajectory. This has been the "holy grail" of DMD gene therapy development. While other developers have struggled to prove that their treatments actually stabilize patients in a way that is measurable and statistically significant, Regenxbio’s data suggests that the presence of their specific microdystrophin protein yields tangible impacts on muscle function.
However, the "mixed" nature of the report cannot be ignored. The occurrence of two serious adverse events (SAEs) has introduced a note of caution into the investment community. Leerink analyst Foroohar noted in an April assessment that these safety signals, while not necessarily disqualifying, complicate the regulatory review. They force the FDA to weigh the potential for life-altering efficacy against the risks of immune-mediated reactions, a calculation that has become increasingly conservative at the agency in the wake of the Elevidys safety incidents.

Official Responses and Strategic Pivot
The leadership at Regenxbio is acutely aware of the "optics" of their situation. CEO Curran Simpson, speaking to analysts during the most recent earnings call, framed the regulatory hurdles not as a roadblock, but as a transitional phase.
"We expect that the new leadership will have a mandate on rare disease flexibility," Simpson stated. "Those are the indications that we’re hearing will be more uniformly adopted, and with that environment, we’re in great shape with our data to push for accelerated approval."
The company’s strategy hinges on a fundamental belief: that the FDA recognizes the desperate, unmet need in the Duchenne community and will prioritize treatments that demonstrate clear biological activity. By delaying the filing, Regenxbio is attempting to avoid the "knee-jerk" regulatory scrutiny that might occur during a period of high turnover, hoping instead to present their case to a more settled, and perhaps more pragmatic, group of regulators in 2027.
Implications: The Future of Rare Disease Regulation
The broader implications of the Regenxbio saga extend far beyond a single drug. The Duchenne gene therapy space has become a bellwether for how the FDA handles the approval of therapies for orphan diseases.
1. The "Surrogate Marker" Dilemma
Regenxbio is banking on the FDA’s willingness to accept microdystrophin expression as a "surrogate marker" for clinical outcomes. If the agency accepts this, it sets a precedent that could accelerate other gene therapy programs. If it rejects this, or demands further confirmatory data, it could signal a return to more traditional, lengthy approval pathways that many biotech firms cannot afford to wait out.

2. The Impact of Agency Turnover
The resignation of Commissioner Marty Makary and the general instability within the FDA’s Center for Biologics Evaluation and Research (CBER) have created a vacuum of predictability. Companies are finding it harder to project timelines, leading to the "counterbalanced" market valuations mentioned by financial analysts. For Regenxbio, the goal is to wait for the storm to pass, but time is a luxury that patients with progressive diseases do not have.
3. Safety vs. Access
The delicate balance between safety and access remains the central tension of the decade for the pharmaceutical industry. The industry is watching to see if the FDA will prioritize the "flexibility" mentioned by Simpson, or if it will lean into a risk-averse stance that prioritizes safety at the expense of potential innovation.
4. Investor Sentiment and Market Viability
The fact that Regenxbio’s share price has struggled despite positive efficacy data reflects a fundamental skepticism among investors. The market is pricing in the "regulatory risk" as heavily as the "commercial opportunity." If Regenxbio can successfully navigate the approval process in 2027, it could restore confidence in the sector. If not, it may serve as further evidence that the commercialization of complex gene therapies for rare diseases is an increasingly precarious business model.
Conclusion
Regenxbio stands at the threshold of a pivotal moment. The data provided for RGX-202 is, by most scientific accounts, encouraging. It offers a tangible mechanism for protein production that has shown a correlation with patient stability. Yet, the company is fighting a battle on two fronts: the clinical challenge of managing safety risks and the geopolitical challenge of navigating a shifting FDA hierarchy.
Whether or not the "mandate for flexibility" that CEO Curran Simpson predicts actually materializes remains to be seen. For the families living with Duchenne, the hope is for a breakthrough that is both safe and effective. For Regenxbio, the next twelve months will be an exercise in patience, strategic communication, and the hope that the regulatory pendulum swings in their favor.