In a significant leap forward for geroscience, researchers at the University of California, Los Angeles (UCLA) have identified a specific subset of "zombie" immune cells that act as a primary engine for tissue degradation, chronic inflammation, and metabolic disease. Published in the journal Nature Aging, the study illuminates how these malfunctioning macrophages accumulate in the body as we age—and, more alarmingly, how they are accelerated by modern, high-cholesterol diets.
The findings offer more than just a biological explanation for liver decline; they present a transformative paradigm for medicine. By selectively purging these senescent cells in mice, researchers were able to reverse severe liver damage and restore metabolic function, even while the animals continued to consume an unhealthy, high-fat diet.
The Mechanism: When Immune Cells Go "Zombie"
At the heart of the research is the phenomenon of cellular senescence. Under normal circumstances, cells that have reached their limit of division or have been damaged by stress undergo a programmed death known as apoptosis. However, in a senescent state, these cells do not die. Instead, they linger in a dormant, dysfunctional state.
Often referred to as "zombie cells," these entities are metabolically active but no longer perform their intended tasks. Worse, they secrete a cocktail of inflammatory proteins and signaling molecules that disrupt the healthy tissue surrounding them.
"Senescent cells are fairly rare, but think of them like a broken-down car on the 405 freeway," explains Anthony Covarrubias, senior author of the study and a member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA. "Just one stalled car can back up traffic for miles. Now imagine five or ten of them slowly accumulating. That’s what these cells do to a tissue: even a small number causes enormous disruption."
Solving the Macrophage Mystery
For decades, the scientific community was divided on whether macrophages—the body’s primary "cleanup crew" responsible for phagocytosis and patrolling for pathogens—could actually become senescent. Because healthy macrophages naturally express some of the markers associated with stress, distinguishing between a hard-working immune cell and a "zombie" cell proved notoriously difficult.
The UCLA team bypassed this hurdle by establishing a definitive molecular signature. They discovered that the presence of two specific proteins, p21 and TREM2, serves as a reliable "fingerprint" for senescent macrophages.
Using this marker, the team observed a startling correlation between age and cellular decline. In the livers of young mice, roughly 5% of macrophages were senescent. In older mice, that number surged to between 60% and 80%. This massive accumulation mirrored the chronic, low-grade inflammation that typically defines the aging process.
The Cholesterol Connection: A Modern Trigger
While aging is a natural driver of this cellular accumulation, the study identifies a more immediate culprit: dietary cholesterol.
"Physiologically, macrophages can handle cholesterol metabolism," notes Ivan Salladay-Perez, the study’s lead author and a graduate student in the Covarrubias lab. "But in a chronic state, it’s pathological. When you look at fatty liver disease, which is driven by overnutrition and too much cholesterol in the blood, that excess cholesterol appears to be a major driver of the senescent macrophage population."
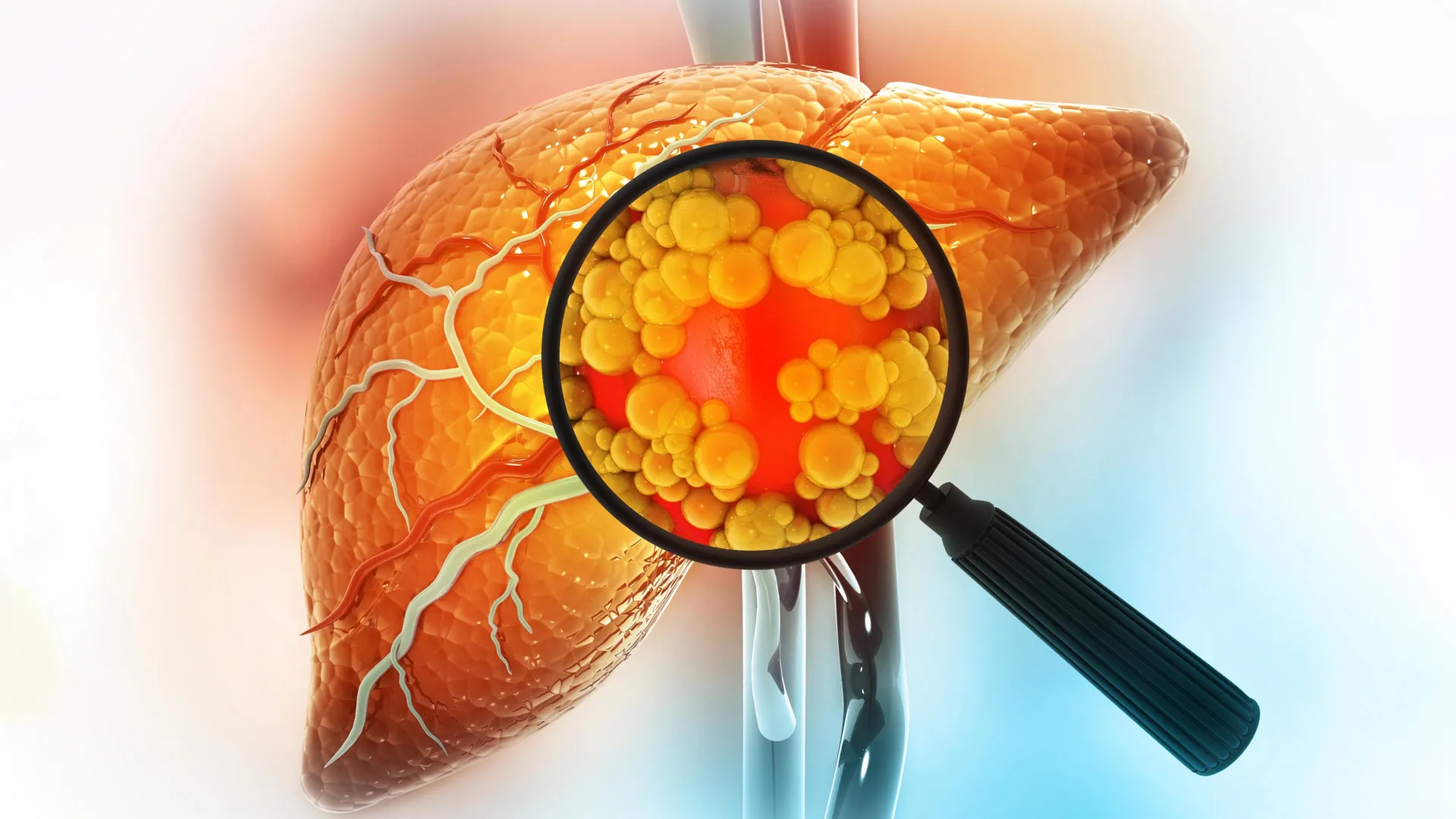
When researchers exposed healthy macrophages to high levels of LDL cholesterol in a laboratory setting, the cells ceased their normal functions, began releasing inflammatory proteins, and adopted the telltale p21-TREM2 molecular signature. This suggests that the modern Western diet—high in processed fats and cholesterol—may be actively "poisoning" our immune system, accelerating biological aging and predisposing organs to chronic disease far earlier than nature intended.
Reversing Damage: The Promise of Senolytics
The most profound outcome of the study occurred when the team applied a therapeutic intervention. Using a drug known as ABT-263—a "senolytic" agent designed to selectively kill senescent cells—the researchers targeted the zombie macrophages in mice fed an unhealthy diet.
The results were transformative:
- Liver Restoration: The livers of the treated mice returned to a healthy weight (decreasing from 7% of body weight to a lean 4–5%).
- Visual Improvement: The organ’s appearance transitioned from an enlarged, yellowish, fatty mass to a healthy, normal red.
- Systemic Impact: The mice experienced a roughly 25% reduction in total body weight, falling from 40 grams to 30 grams, without any modifications to their caloric intake.
"That’s what wowed me," Salladay-Perez said. "Eliminating senescent cells doesn’t just slow the fatty liver—it actually reverses it."
Clinical Implications: A Looming Public Health Crisis
The research is not confined to rodent models. By analyzing human liver biopsy datasets, the UCLA team confirmed that the p21-TREM2 signature is significantly elevated in human patients with advanced liver disease.
This is a critical concern, particularly in metropolitan areas like Los Angeles. Estimates suggest that 30% to 40% of the local population is affected by fatty liver disease, with disproportionately high rates in Latino communities. Current treatment options are largely limited to lifestyle interventions, which can be difficult to sustain and ineffective for those with advanced damage.
"This is a huge public health crisis in the making," says Covarrubias. "We’re seeing fatty liver disease in younger and younger people. We are really happy to make some inroads into understanding what’s driving it and identifying cell types we might be able to target."
Future Directions: Beyond the Liver
While the results of the study are encouraging, the team emphasizes that ABT-263 is too toxic for human clinical use due to its broad side effects. The current focus of the lab has shifted to identifying safer, more targeted compounds that can clear senescent macrophages without harming healthy tissue.
The scope of this research is also expanding. The team is currently investigating whether similar processes occur in the brain. Microglia, the macrophages of the central nervous system, are known to encounter massive amounts of cellular debris. If these cells become senescent, they could potentially contribute to neurodegenerative conditions such as Alzheimer’s disease.
This aligns with the Geroscience Hypothesis, a growing field of study that posits that the underlying mechanisms of aging—such as the buildup of senescent cells—are the root causes of most chronic diseases. If scientists can master the ability to clear these cells, they may eventually be able to treat not just fatty liver disease, but atherosclerosis, cancer, and dementia as part of a single, unified medical strategy.
"If you really understand the basic mechanisms driving inflammation with aging, you can target those same mechanisms to treat not just fatty liver disease, but atherosclerosis, Alzheimer’s, and cancer," Salladay-Perez concluded. "It all goes back to understanding how these cells arise in the first place."
As the scientific community continues to explore the "zombie cell" phenomenon, the promise of a future where we can surgically remove the cellular remnants of aging becomes increasingly tangible, offering hope for a longer, healthier human lifespan.
The study was supported by the National Institutes of Health, the Glenn Foundation for Medical Research, the American Federation for Aging Research, and the UCLA-UCSD Diabetes Research Center.