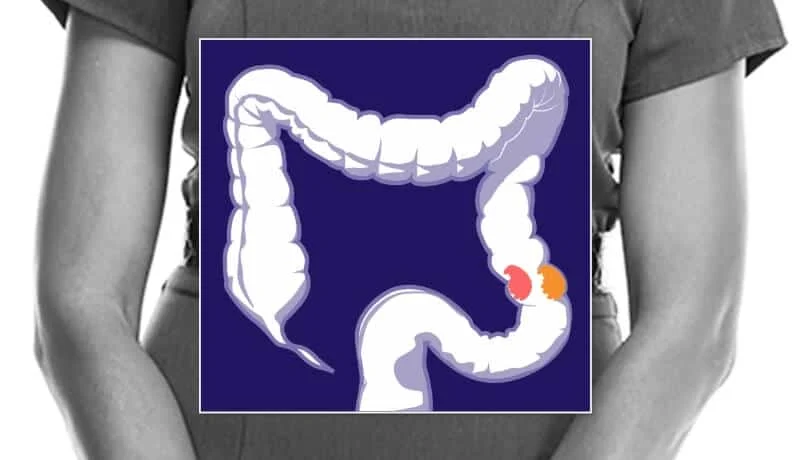
Colorectal cancer (CRC) is currently undergoing a paradoxical shift in the landscape of oncology. Once considered a disease primarily affecting the elderly, it has rapidly ascended to become the second leading cause of cancer-related death in the United States and the number one killer among adults under 50. Despite the transformative potential of immunotherapy—a medical breakthrough that has turned once-terminal diagnoses into stories of long-term remission for melanoma and lung cancer patients—it remains largely ineffective for the vast majority of those battling colorectal tumors.
Dr. Karin Pelka, an investigator at the Gladstone Institutes and a recipient of the prestigious Cancer Research Institute (CRI) Technology Impact Award, is spearheading a bold initiative to bridge this gap. Her research does not merely look at the tumor as a static mass; it seeks to "eavesdrop" on the complex molecular conversations occurring within the tumor microenvironment (TME) to unlock the immune system’s latent power.
The Main Facts: Why Colorectal Cancer Defies Modern Treatment
The clinical challenge of colorectal cancer lies in its immunological "invisibility." For immunotherapy—specifically checkpoint inhibitors—to work, the immune system must first recognize the tumor as a foreign intruder. In cancers like melanoma, high rates of DNA mutation create "neoantigens," or molecular flags, that signal the immune system to attack.
However, most colorectal cancers possess a different genetic profile. They are often "cold" tumors, meaning they do not trigger a robust immune response, leaving the body’s natural defenses blind to the encroaching disease.
The dMMR Exception
There is a critical subset of patients—roughly 5% to 10%—whose tumors exhibit DNA mismatch repair (dMMR) defects. Because these tumors are hyper-mutated, they are highly visible to the immune system. In a landmark 2022 clinical trial, patients with locally advanced rectal cancer carrying these defects saw a 100% response rate to immunotherapy alone, effectively eliminating the need for toxic chemotherapy, radiation, or disfiguring surgery. Dr. Pelka’s work is focused on the fundamental question: How can we replicate this success for the 90% of patients who do not carry these specific genetic markers?
Chronology of a Crisis and a Scientific Pivot
The Rise of Early-Onset Colorectal Cancer
The urgency of this research is underscored by epidemiological data showing a disturbing surge in diagnoses among adults under 50. While the exact causes remain a subject of intense study—ranging from dietary shifts to environmental factors—the biological behavior of these "early-onset" tumors often appears more aggressive and resistant to standard care.
- 1990s–2010s: The era of chemotherapy and standard surgical intervention. While survival rates improved, they plateaued for metastatic cases.
- 2017: The FDA approves immunotherapy (pembrolizumab) for dMMR/MSI-H solid tumors, marking the first time a treatment was approved based on a genetic marker rather than the site of the cancer.
- 2022: The NEJM study confirms that immunotherapy can serve as a primary treatment for specific rectal cancers, effectively replacing standard-of-care protocols.
- 2025–2026: The current focus shifts to "cold" tumors. Researchers like Dr. Pelka begin utilizing spatial transcriptomics and AI to understand why the immune system fails to activate in the vast majority of CRC patients.
Mapping "Immune Hubs": A New Understanding of Tumor Dynamics
Dr. Pelka’s laboratory has identified a structural phenomenon within tumors that may hold the key to universal immunotherapy success: "immune hubs."
What are Immune Hubs?
Within the chaotic architecture of a tumor, Dr. Pelka discovered organized clusters where killer T cells (the immune system’s soldiers) congregate alongside cancer cells and structural connective tissue. Within these hubs, there is a constant, intense molecular signaling exchange.
The research indicates that the state of these hubs is predictive of patient outcomes. When these hubs are in an "activated" state, they facilitate an immune attack. When they are dormant or dysregulated, the tumor continues to grow unchecked. Crucially, these hubs are not unique to colorectal cancer; they have been observed in lung cancer and melanoma as well. This suggests that the biological "machinery" of immune activation is conserved across cancer types, providing a universal target for future therapeutics.
Teaching AI to Read the Language of the Cell
To map these interactions, Dr. Pelka is utilizing Geneformer, a foundational AI model developed by Dr. Christina Theodoris. If a tumor is a conversation, Geneformer is the linguist capable of deciphering it.
The Role of Foundational AI
Geneformer was trained on tens of millions of human cell profiles, learning the "grammar" of gene expression. By applying this to colorectal cancer data, Dr. Pelka’s team is identifying "central regulators"—the master molecular switches that dictate whether a cancer cell remains invisible to the immune system or broadcasts its presence.
This approach moves beyond simple genetic sequencing. By using spatial transcriptomics, the lab can map exactly where these interactions occur within the tumor’s physical landscape. This creates a high-resolution map of the tumor’s defenses, allowing researchers to pinpoint the exact moment when immune activity is suppressed and intervene with targeted, precision therapies.
Supporting Data and The "Technology Impact"
The CRI Technology Impact Award is designed to fund high-risk, high-reward research that traditional funding agencies might deem too speculative. As Dr. Pelka notes, the transition from basic laboratory discovery to clinical application is often hindered by a "valley of death" in funding.
The Need for Early-Stage Investment
"The CRI Technology Impact Award is a perfect example of bringing together new technologies with the problem of getting cancer immunotherapy to work," Dr. Pelka explains. "Having a foundation willing to take a bet early on—when the science is not yet ready to be employed widely, but where early discoveries can really move the needle—that is often research that is hard to fund, because it is very risky."
The potential impact of this research is backed by the success of earlier immunotherapy breakthroughs. In trials where immune activity was successfully modulated, patients achieved outcomes that were previously deemed impossible. By decoding the "central regulators" of these immune hubs, Dr. Pelka aims to transform the 90% of currently non-responsive patients into the next generation of responders.
Implications: The Future of Patient Care
The ultimate goal of this research is to create a "living therapeutic." Unlike traditional drugs, which the body metabolizes and eliminates, immunotherapy aims to train the immune system to recognize and eliminate cancer cells on an ongoing basis.
Prevention and Early Intervention
While the research focuses on treating advanced disease, the scientific community remains emphatic about the importance of screening. Colorectal cancer is unique in that it is largely preventable. A colonoscopy can identify and remove precancerous polyps before they evolve into malignancies—a process that typically takes 10 to 15 years. Regular screening starting at age 45 remains the most effective tool in the oncologist’s arsenal.
A Note of Hope for Younger Patients
For the growing demographic of young adults diagnosed with CRC, time is the most precious resource. Targeted therapies, while initially effective, often lose their efficacy as cancer cells evolve and develop resistance. Immunotherapy offers a different path: an adaptive response. By "teaching" the immune system to adapt alongside the cancer, researchers are looking toward a future where "cured" is no longer a temporary state, but a long-term reality.
Dr. Pelka’s work represents a fundamental shift from treating the tumor to empowering the host. By decoding the molecular conversations within the immune hubs, she is not just working toward a new drug—she is working toward a permanent change in how we perceive, treat, and ultimately survive colorectal cancer. As the research matures, the hope is that these "master switches" will provide the key to unlocking the full potential of immunotherapy for every patient, regardless of their tumor’s genetic profile.