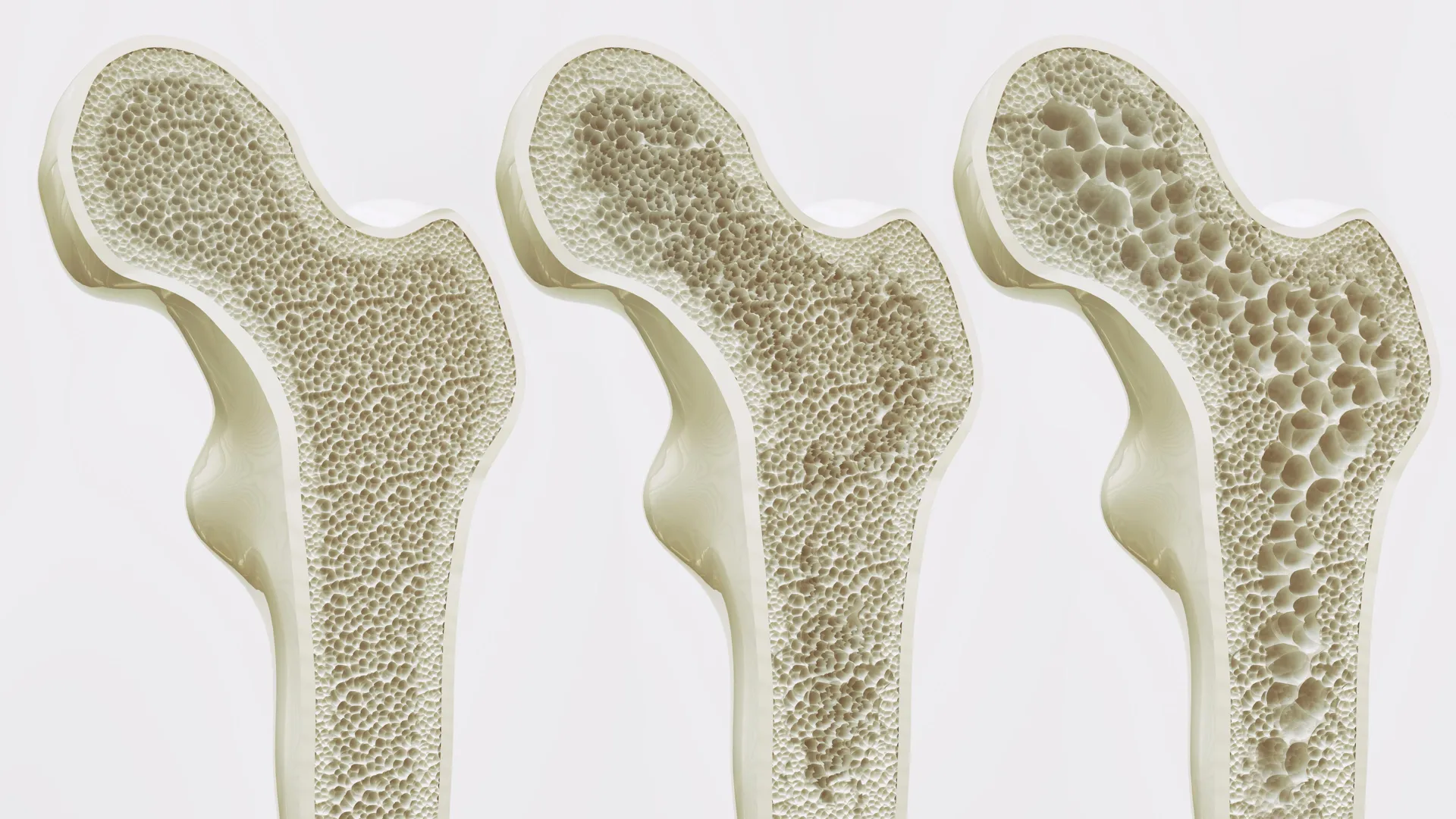
For approximately six million people in Germany alone—and hundreds of millions worldwide—the phrase "silent disease" carries a heavy weight. Osteoporosis, a condition characterized by the gradual thinning and weakening of bone tissue, often remains undetected until the catastrophic moment of a fracture. As global populations age, the demand for medical interventions that do not merely slow bone degradation but actively reverse it has reached a critical juncture.
Now, a team of researchers at Leipzig University has unveiled a potential turning point in bone biology. By identifying a specific biological "switch" known as the GPR133 receptor, scientists have opened the door to a new generation of regenerative therapies. This discovery, rooted in over a decade of rigorous biochemical exploration, suggests that we may soon be able to orchestrate the body’s own internal machinery to rebuild the skeletal structure.
The Biological Foundation: Why Bones Fail
To understand the significance of this discovery, one must first appreciate that bone is not a static architectural element of the body. It is a living, dynamic tissue in a state of perpetual renovation. This process, known as bone remodeling, is governed by a delicate equilibrium between two primary cell types: osteoblasts, which act as the body’s "masons" to construct new bone tissue, and osteoclasts, the "demolition crew" that breaks down old or damaged bone.
In healthy individuals, these processes are perfectly synchronized. However, with the onset of aging and the hormonal shifts associated with menopause, this balance is disrupted. Osteoclast activity frequently outpaces that of osteoblasts, leading to a net loss of bone density. Current pharmacological treatments, such as bisphosphonates, largely focus on slowing the "demolition" side of this equation. While effective, they often carry limitations and side effects, leaving a clinical void for treatments that can actively stimulate the construction of new, robust bone.
The Discovery of GPR133: A New Biological Target
The breakthrough from the Rudolf Schönheimer Institute of Biochemistry centers on a protein known as GPR133. This receptor belongs to the Adhesion G protein-coupled receptor (aGPCR) family—a group of surface proteins that facilitate communication between cells and their environment. While GPCRs have long been the target of existing medicines for various conditions, the adhesion subgroup has historically remained a "dark corner" of pharmacology, largely misunderstood and under-researched.
Leipzig University’s Collaborative Research Centre 1423, which focuses on the structural dynamics of GPCR activation, has spent the last ten years mapping these receptors. Their research has now established that GPR133 is not merely a bystander; it is a key regulator of bone integrity.
When activated, GPR133 acts as a signaling conductor. It senses environmental cues—such as mechanical pressure and interactions between neighboring cells—and translates them into a biological command: "Increase osteoblast activity, decrease osteoclast activity." By tipping the scales in favor of bone formation, GPR133 serves as a natural guardian of skeletal strength.
Chronology of the Research
The path to this discovery was not linear; it was the result of a long-term, multidisciplinary commitment to structural biochemistry.
- 2014–2020: Mapping the Unknown: Researchers at Leipzig University began an intensive study of adhesion GPCRs under the umbrella of the Collaborative Research Centre 1423. The goal was to decode how these receptors function at a molecular level.
- 2021: Identifying the Target: Through advanced genetic modeling, the team observed that mice lacking the GPR133 receptor exhibited premature bone loss. These animal models mirrored the clinical progression of human osteoporosis, providing a clear causal link between the receptor and bone health.
- 2022: Computational Screening: To find a way to manipulate the receptor, the team turned to computer-assisted drug discovery. They screened vast libraries of molecules to identify candidates capable of stimulating GPR133. This process led to the identification of AP503.
- 2023–2024: Validation: In controlled trials, the administration of AP503 to both healthy and osteoporotic mice resulted in a significant increase in bone mineral density and overall bone strength.
Official Perspectives: Translating Science into Medicine
The lead investigators behind the study emphasize that the findings are more than just a theoretical success; they represent a potential shift in the therapeutic paradigm.
"If this receptor is impaired by genetic changes, mice show signs of loss of bone density at an early age—similar to osteoporosis in humans," explains Professor Ines Liebscher, the study’s lead investigator. "Using the substance AP503, which was identified via a computer-assisted screen as a stimulator of GPR133, we were able to significantly increase bone strength in both healthy and osteoporotic mice."
The clinical potential is further amplified by the receptor’s versatility. Dr. Juliane Lehmann, the lead author of the study, notes that the impact of GPR133 is not isolated to the skeleton. Previous findings by the same team indicated that activating GPR133 also bolsters skeletal muscle strength.
"The newly demonstrated parallel strengthening of bone once again highlights the great potential this receptor holds for medical applications in an aging population," says Dr. Lehmann. For the elderly, the dual benefit of bone and muscle reinforcement is the "holy grail" of fall prevention—a crucial factor in maintaining independence and reducing fracture-related mortality.
Implications for an Aging Society
The broader implications of the GPR133 research are profound. As the global population shifts toward an older demographic, the societal and economic burden of osteoporosis—manifesting in hip, spine, and wrist fractures—is projected to climb.
A Two-Pronged Approach
Most current treatments for osteoporosis are "anti-resorptive," meaning they aim to prevent further loss. The promise of AP503 and future GPR133-targeted therapies lies in their "anabolic" potential—the ability to stimulate the growth of new bone. This shift from damage control to restorative care could change the prognosis for patients who have already experienced significant bone density loss.
The Muscle-Bone Crosstalk
The discovery also highlights the importance of "crosstalk" between muscle and bone. By targeting a receptor that influences both systems, researchers may be able to create a singular therapy that addresses the fragility of the musculoskeletal system as a whole. This is particularly relevant for the treatment of sarcopenia (muscle loss) and osteoporosis simultaneously, a common comorbid condition in the elderly known as "osteosarcopenia."
Looking Ahead: The Road to Human Trials
While the results in murine models are highly encouraging, the research team remains cautious and diligent. The transition from bench to bedside is a rigorous process. Future phases of the research will focus on:
- Safety and Toxicology: Ensuring that the systemic activation of GPR133 through compounds like AP503 does not produce unintended off-target effects in other tissues.
- Pharmacokinetics: Determining the optimal delivery method and dosage to maximize therapeutic benefit in humans.
- Clinical Trials: Designing robust human trials to confirm the efficacy of AP503 in reversing bone loss in post-menopausal women and men with age-related osteoporosis.
The work of the Leipzig team, supported by the Collaborative Research Centre 1423, serves as a testament to the importance of basic science. By focusing on the structural dynamics of GPCRs, they have unlocked a pathway that was previously invisible to the medical community.
As the researchers continue to explore the functions of GPR133 throughout the body, the scientific community watches with anticipation. If these findings hold true in human trials, the future of osteoporosis treatment may no longer be defined by the management of decline, but by the potential for renewal. The "silent disease" may finally be finding a voice—and a cure—in the form of targeted molecular therapy.