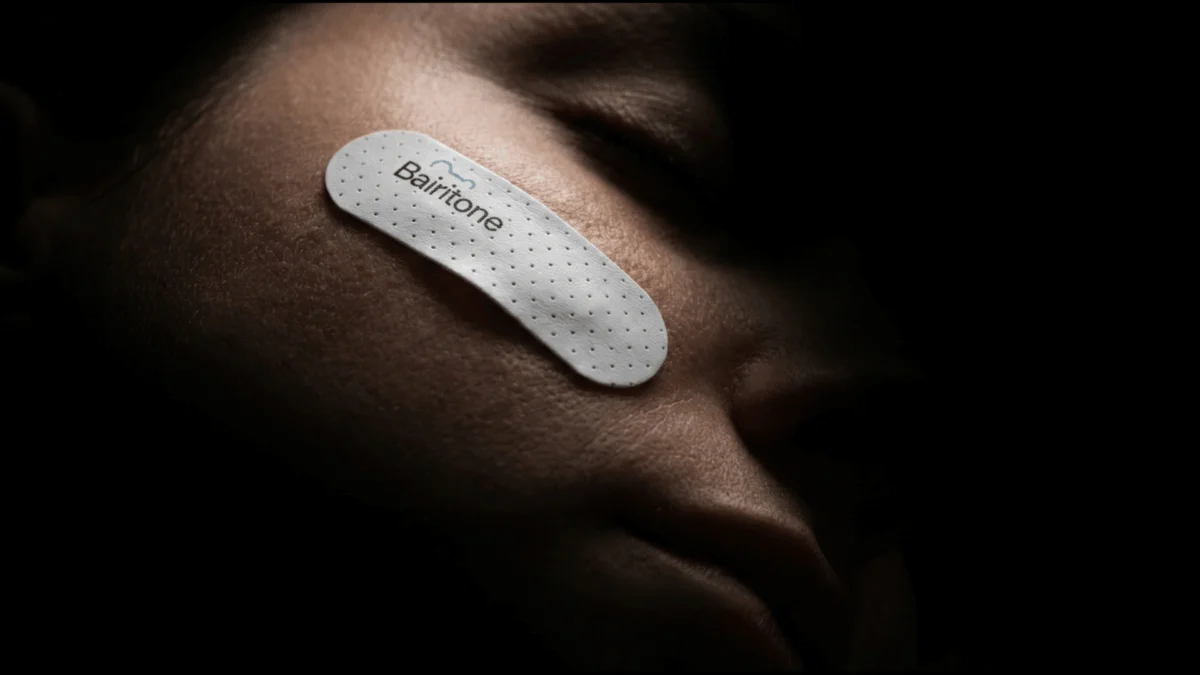
In the landscape of modern sleep medicine, the diagnosis of Obstructive Sleep Apnea (OSA) has long remained a cumbersome, often binary process. For millions of sufferers, the standard diagnostic pathway—typically involving a polysomnography study in a clinical setting—often reveals the existence of an obstruction without effectively identifying its precise anatomical origin. However, a significant paradigm shift may be on the horizon. Bairitone Health, a medical technology innovator, recently announced that its proprietary sleep sonar platform has been granted Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA). This milestone marks a pivotal moment for a company aiming to replace invasive, static diagnostic imagery with dynamic, non-invasive anatomical mapping during natural sleep.
The FDA Breakthrough Designation: A Seal of Clinical Urgency
The FDA’s Breakthrough Device Designation is not merely a bureaucratic checkbox; it is a rigorous recognition reserved for medical devices that offer the potential to provide more effective treatment or diagnosis of life-threatening or irreversibly debilitating human conditions. By entering this program, Bairitone Health gains prioritized access to FDA experts, enabling a streamlined premarket review process.
For the medical community, this designation serves as a strong signal that current OSA diagnostic methodologies are insufficient. Obstructive sleep apnea is not a monolithic condition; it is a complex, multi-factorial disorder that often leaves clinicians guessing whether a patient’s airway collapse is occurring at the level of the palate, the tongue base, or the epiglottis. The FDA’s involvement suggests that Bairitone’s approach—utilizing passive sleep sonar—addresses a significant "unmet need" in the standard of care, offering a pathway toward precision medicine in a field often characterized by "one-size-fits-all" CPAP (Continuous Positive Airway Pressure) therapy.
The Technology: Decoding the Anatomy of a Breath
At the core of Bairitone Health’s innovation is a departure from traditional imaging modalities such as MRI, X-ray, or ultrasound. These traditional methods, while valuable, capture a "snapshot" of the airway. Because they often require the patient to be awake or in a controlled environment, they fail to replicate the complex, shifting dynamics of the human airway during the various stages of natural sleep.
Bairitone’s platform functions through a sophisticated integration of a discrete facial patch and passive sleep sonar technology. By mapping airway vibrations, the system effectively "listens" to the anatomy as the patient sleeps. This dynamic approach allows the technology to track airway behavior across different sleep stages, body positions, and breathing patterns.
Key Technical Capabilities:
- Multi-level Obstruction Profiling: Identifying whether a patient suffers from single or multiple points of collapse within the upper airway.
- Primary vs. Secondary Collapse Identification: Distinguishing between the root cause of the obstruction and the subsequent physiological effects.
- Flow-Limitation Detection: Pinpointing the exact anatomical sites where airflow is restricted, even before a full collapse occurs.
- Phenotyping and Selection: Assisting clinicians in selecting candidates for specific surgical or non-surgical interventions.
- Therapy Titration Optimization: Providing data that explains why certain therapies may be failing, allowing for adjustments that improve patient outcomes.
Chronology: The Evolution of Bairitone’s Vision
The journey to this FDA milestone is rooted in the broader evolution of sleep technology. For decades, the gold standard for OSA diagnosis—the In-Laboratory Polysomnography (PSG)—has provided essential data regarding oxygen saturation, heart rate, and sleep staging. However, it has historically lacked the resolution to visualize the why behind the what.
- Initial Research Phase: Bairitone Health began with the premise that airway vibrations contain a wealth of untapped data. By shifting the focus from simply detecting apnea events to understanding the acoustic signature of the airway, the company developed the foundational sonar algorithms.
- Validation Studies: Through internal trials, the company demonstrated that its passive sonar could accurately correlate acoustic data with known anatomical structures, effectively mapping the upper airway without the need for radiation or confinement in an imaging suite.
- FDA Engagement: Recognizing the potential to revolutionize sleep medicine, Bairitone initiated the rigorous application process for the Breakthrough Device Program. The successful designation underscores the robustness of the data presented to regulators and the potential for the technology to disrupt existing diagnostic norms.
Official Perspectives: The Leadership Vision
Dr. Meagan Pitcher, CEO and co-founder of Bairitone Health, views the FDA designation as a validation of the company’s patient-centric philosophy. "At Bairitone, we’re honored to receive FDA Breakthrough Device Designation for our noninvasive diagnostic technology in obstructive sleep apnea," Dr. Pitcher stated in a recent press release.
Her sentiments highlight a central challenge in sleep medicine: the disconnect between diagnostic data and actionable therapy. Dr. Pitcher emphasizes that the current diagnostic landscape often leaves clinicians without a clear roadmap for treatment. By bringing "precise anatomical insight" into the equation, Bairitone aims to reduce the trial-and-error approach that often frustrates OSA patients. The goal, according to the leadership team, is to make advanced diagnostic data both "more accessible and more practical" for the clinicians who manage the day-to-day care of patients.
Implications for the Future of Sleep Medicine
The implications of Bairitone’s platform extend far beyond the laboratory. If successfully cleared for clinical use, the technology could fundamentally change the treatment algorithm for OSA.
1. The Death of Trial-and-Error Therapy
Currently, many patients are prescribed CPAP therapy as a first-line treatment. While highly effective for many, adherence rates are notoriously low, and for some, CPAP is simply not the correct solution for their specific anatomical obstruction. By providing a "map" of the airway, Bairitone’s technology could allow physicians to identify early on which patients might benefit more from oral appliance therapy, hypoglossal nerve stimulation, or targeted surgical interventions, thereby increasing the likelihood of successful treatment on the first attempt.
2. Democratizing Access to Specialized Care
Advanced airway analysis often requires referral to specialized ENT (Ear, Nose, and Throat) surgeons or sleep specialists with access to sophisticated imaging equipment. A non-invasive, wearable patch-based system could potentially be deployed in a wider variety of clinical settings, including primary care, thereby reducing the time to accurate diagnosis and significantly decreasing the burden on the healthcare system.
3. Precision Medicine in Sleep
The ability to phenotype patients—to categorize them based on the specific mechanical cause of their OSA—is the hallmark of modern precision medicine. Bairitone’s platform provides the data necessary to transition from treating "sleep apnea" as a single diagnosis to treating specific "obstructive phenotypes." This granularity is expected to improve not only patient comfort but also long-term cardiovascular and metabolic outcomes, which are closely linked to untreated OSA.
Supporting Data and Technical Nuance
While Bairitone’s platform is currently for investigational use only, the scientific foundation relies on the acoustic properties of the airway. The human pharynx acts as a resonant chamber. During sleep, the soft tissues of the airway lose muscle tone, leading to varying degrees of narrowing. This narrowing alters the acoustic signature of the air passing through the pharynx. By utilizing high-fidelity sensors, Bairitone’s sonar captures these minute changes in frequency and amplitude, translating them into a three-dimensional model of the airway’s functional state.
This data is particularly powerful when analyzed across the "sleep architecture." By observing how the airway responds to the transition from NREM to REM sleep—where muscle tone is significantly reduced—the system can identify "weak links" in the anatomy that might be missed during a stationary or awake imaging exam.
Addressing the Unmet Needs
The FDA’s decision to grant Breakthrough status serves as a reminder that the status quo is failing a significant portion of the population. Obstructive Sleep Apnea is not merely a nuisance; it is a major risk factor for hypertension, stroke, type 2 diabetes, and cognitive decline. The "unmet need" cited by the FDA refers to the lack of tools that can effectively guide personalized therapy.
Bairitone Health’s platform seeks to fill this void by providing:
- Reduced Patient Burden: No claustrophobic MRI machines or radiation exposure.
- Increased Clinical Efficiency: Providing actionable data that reduces the need for repeated, ineffective follow-up tests.
- Evidence-Based Decisions: Replacing anecdotal treatment choices with clear, objective anatomical data.
Conclusion: A Turning Point for OSA Patients
As Bairitone Health moves forward through the premarket review phase, the medical community will be watching closely. The prospect of an "anatomical map" for the sleeping airway represents a significant leap forward in our understanding of sleep-disordered breathing. While the technology is still pending final clearance, the FDA’s endorsement provides a strong foundation of confidence.
For the millions of patients currently struggling with the discomfort of CPAP or the uncertainty of an undiagnosed obstruction, the work being done at Bairitone represents more than just a new piece of hardware; it represents the potential for a new era of personalized sleep medicine. By turning the lights on the "silent night" of the airway, Bairitone Health is helping ensure that every patient can finally breathe—and sleep—more easily.