In the high-stakes battlefield of the human body, the cytotoxic T lymphocyte (CTL) acts as a highly specialized surgical strike force. These immune cells are the body’s primary defense against malignant tumors and viral pathogens, tasked with the lethal responsibility of identifying, engaging, and destroying compromised cells while sparing the healthy neighbors that surround them.
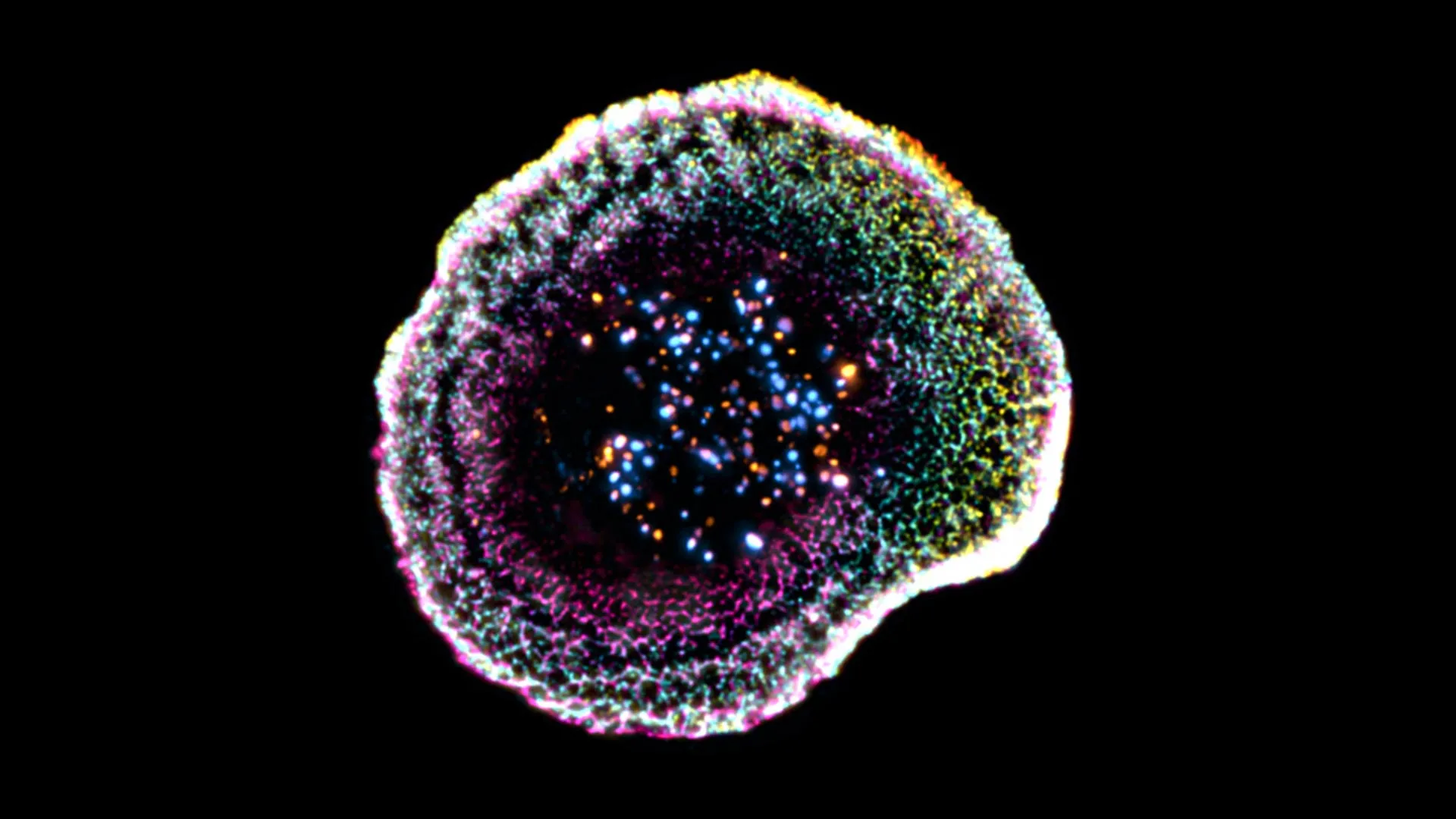
For decades, the mechanics of this process have been well-understood in theory, yet the physical "machinery" of the interaction—the immune synapse—has remained notoriously difficult to observe in its natural state. Now, a groundbreaking collaboration between the University of Geneva (UNIGE) and the Lausanne University Hospital (CHUV) has shattered these observational barriers. By utilizing a cutting-edge technique known as cryo-expansion microscopy (cryo-ExM), researchers have captured the first high-resolution, three-dimensional look at these cells in near-native conditions. Their findings, recently published in the journal Cell Reports, represent a paradigm shift in how we visualize the frontline of human immunity.
The Immune Synapse: A Lesson in Biological Precision
At the heart of the cytotoxic T cell’s effectiveness is the "immune synapse." When a T cell detects a target—be it a cell hijacked by a virus or a cell undergoing cancerous transformation—it does not simply release toxins into the bloodstream. Instead, it docks onto the target, forming a tight, specialized junction.
This synapse acts as a highly regulated conduit. By focusing the release of toxic proteins—such as perforin and granzymes—directly into the target cell, the T cell ensures a rapid, decisive strike. This "targeted delivery" mechanism is essential; indiscriminate chemical warfare would lead to massive collateral damage, destroying healthy tissues and causing systemic inflammation. However, until this study, the exact internal structural dynamics that allow the T cell to form this seal and calibrate its weapon systems remained obscured by the limitations of traditional microscopy.
Chronology of Discovery: Overcoming the Imaging Barrier
The history of cellular imaging has long been a struggle against the physics of light and the fragility of biological matter. Historically, researchers faced an agonizing trade-off: they could achieve high resolution, but only by using harsh chemical fixatives that warped the cell’s internal architecture. Or, they could use live-cell imaging, which offered a window into movement but lacked the nanometer-scale detail necessary to understand the structural proteins at play.
The Evolution of the Method
The road to the current discovery began with the refinement of expansion microscopy, a technique that involves physically enlarging a biological sample to make it easier to image. However, standard expansion techniques often compromised the integrity of delicate intracellular structures.
- Phase One: Vitrification. The UNIGE and CHUV team, supported by the ISREC Foundation TANDEM program, pivoted to cryo-expansion microscopy. The process begins with "vitrification"—an instantaneous freezing technique that turns water into a glass-like solid rather than ice crystals, which would otherwise puncture and distort the cell’s organelles.
- Phase Two: Expansion. Once vitrified, the sample is infused with an absorbent hydrogel. When triggered, the hydrogel expands, pulling the molecular components of the cell apart in a perfectly uniform manner. This allows standard microscopes to resolve details that were previously far below the diffraction limit of light.
- Phase Three: Visualization. By applying this to T cells interacting with target cells, the team was able to map the spatial organization of the synapse in 3D, providing a "high-definition" view of the internal architecture that supports the cell’s killing function.
Unveiling the Nanoscale: New Structural Insights
The study has revealed several previously hidden features that define the T cell’s combat readiness. Among the most significant findings is the discovery of the "dome" structure at the point of contact.
The Dome Effect
"Our work reveals that at the point of contact between the immune cell and its target, the membrane forms a kind of dome, whose structure appears to be linked to adhesion interactions and to the internal organization of the cell," explains Florent Lemaître, a postdoctoral researcher at UNIGE and the study’s lead author. This dome appears to be more than just a byproduct of physical contact; it is an organized, reinforced structural interface that likely stabilizes the synapse, ensuring the T cell does not detach prematurely during the chemical discharge.
The Complexity of Cytotoxic Granules
The research team also provided the first clear look at the internal variability of cytotoxic granules—the "ammunition" storage units within the T cell. Previously, these were often treated as uniform packages of toxins. The cryo-ExM imagery shows that these granules possess internal heterogeneity. Some contain a single dense core of toxic material, while others contain multiple cores. This variation suggests that T cells may be able to calibrate the "payload" of their strike depending on the nature or the resilience of the target cell.
From Petri Dish to Patient: Clinical Implications
Perhaps the most ambitious aspect of the study was the transition from lab-grown cells to human tumor samples. The ability to observe these mechanisms in clinical tissue—where the environment is cluttered with non-immune cells, connective tissue, and extracellular matrix—is a significant leap forward.
Bridging the Gap
"We extended this approach to human tumor tissues, making it possible to directly observe T lymphocytes infiltrating tumors and their cytotoxic machinery at the nanometer scale," says Dr. Benita Wolf, Chief Resident and Associate Researcher at CHUV, who co-led the study.
By seeing these interactions within the complex, hostile environment of a tumor, researchers can now ask questions that were previously impossible to address:
- Why do some T cells fail to dock with tumor cells?
- How does the tumor microenvironment physically prevent the formation of the synapse?
- Can we structurally reinforce the immune synapse to make current immunotherapies more effective?
This clinical application allows for the study of the immune response in its "real-world" context, potentially revealing why certain tumors are resistant to immunotherapy. If the T cell cannot form the structural "dome" or if its cytotoxic granules are failing to reach the synapse, the treatment will inevitably fail. Knowing exactly where the breakdown occurs provides a roadmap for future drug development.
Official Responses and Future Outlook
The scientific community has reacted with cautious optimism, viewing this as a tool that could revitalize stagnant areas of immuno-oncology. The ISREC Foundation, which backed the TANDEM program, emphasized that the collaboration between basic scientists at UNIGE and clinicians at CHUV was the critical catalyst for this success.
"The work offers a valuable framework for studying immune responses in real-world conditions," the authors noted in their discussion. By providing a 3D view of the T cell’s internal machinery, the researchers have effectively opened a "black box" that has hindered cancer research for decades.
Future Directions
The team plans to use this imaging platform to screen how different cancer drugs affect the structure of the immune synapse. If a certain chemotherapy drug, for instance, is found to disrupt the formation of the synaptic dome, it could explain why that drug might interfere with the efficacy of immune-checkpoint inhibitors.
Furthermore, the methodology of cryo-expansion microscopy is not limited to T cells. It can be applied to any biological system where the relationship between structure and function is paramount—from neurons in the brain to the viral replication cycles within a host cell.
Conclusion: A New Era of Visualizing Immunity
As we enter an era where cancer treatment is increasingly defined by our ability to harness the body’s own defenses, the need for high-fidelity data has never been greater. The UNIGE-CHUV study serves as a stark reminder that the immune system is a master of physical engineering. By successfully mapping the nanoscale architecture of the killer T cell, researchers have moved closer to a future where we can not only stimulate the immune system but also repair its physical shortcomings.
This study does more than just present beautiful, high-resolution images; it provides a new set of eyes for oncologists and immunologists alike. As the field moves forward, the "dome" of the immune synapse and the "cores" of the cytotoxic granules may well become the new targets for the next generation of precision medicine, turning the tide in the ongoing struggle against cancer.