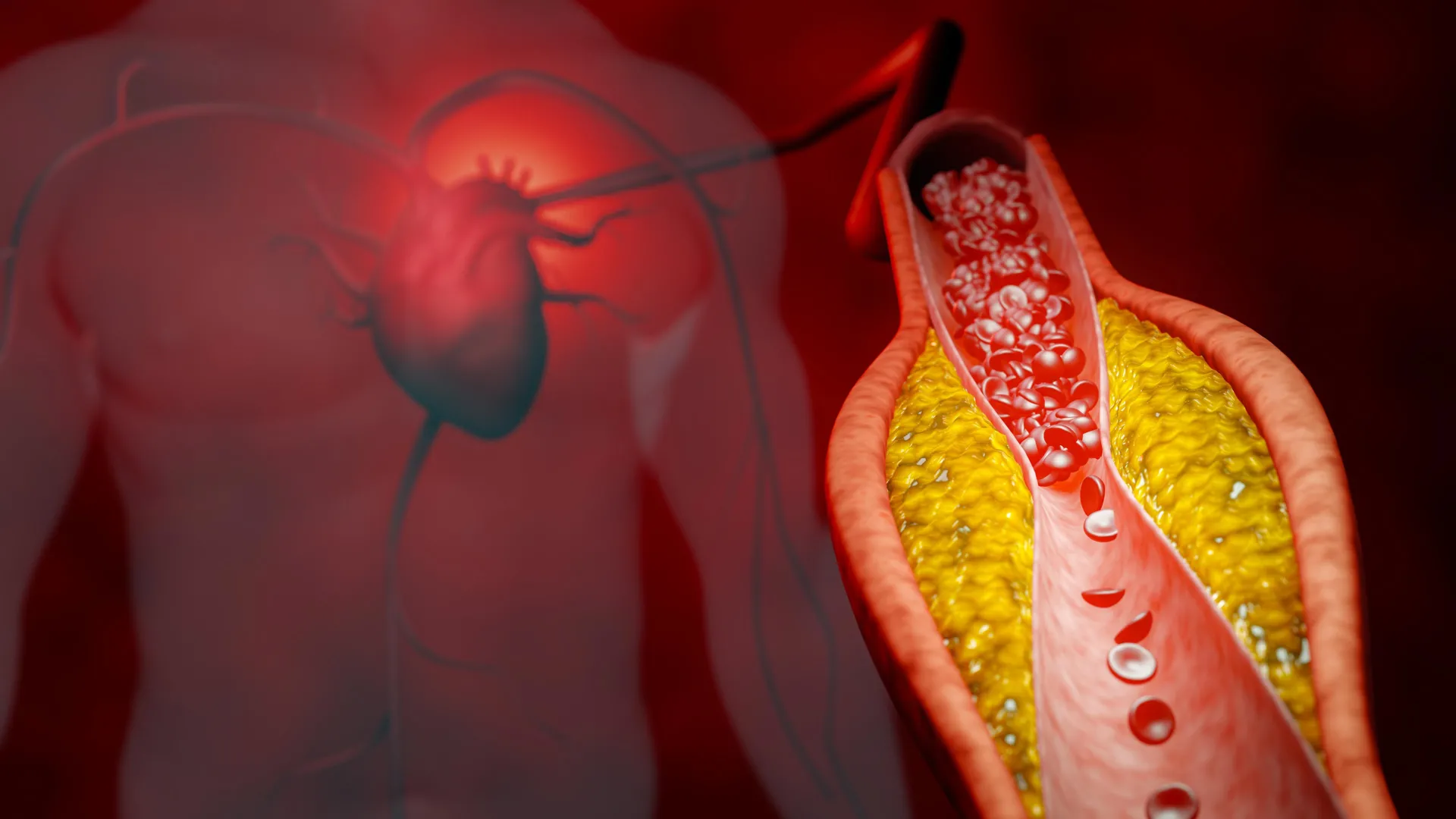
High levels of cholesterol in the bloodstream—clinically referred to as hypercholesterolemia—represent one of the most pervasive health challenges of the modern era. As a primary driver of atherosclerosis, the process by which fatty plaques calcify and constrict arterial walls, this condition remains a leading precursor to heart attacks, strokes, and peripheral vascular disease. While pharmaceutical interventions like statins have dominated the landscape of cardiovascular care for decades, they are often accompanied by significant side effects, including muscle pain (myopathy) and potential metabolic shifts.
Now, a pioneering collaboration between the University of Barcelona and the University of Oregon has introduced a paradigm-shifting approach. By utilizing specialized DNA-based molecules known as polypurine hairpins (PPRHs), researchers have successfully developed a method to suppress the protein PCSK9 at the genetic level. This breakthrough, recently published in the journal Biochemical Pharmacology, offers a potentially safer, more precise, and highly effective alternative to current cholesterol-lowering regimens.
The Main Facts: Silencing the Protein Behind High Cholesterol
At the heart of this research is the protein PCSK9 (proprotein convertase subtilisin/kexin type 9). In a healthy biological system, low-density lipoprotein (LDL) receptors on the surface of liver cells act as "gatekeepers," scavenging "bad" cholesterol from the bloodstream to keep levels in check. However, PCSK9 acts as a biological antagonist to these receptors. It binds to the LDL receptors and encourages their degradation, effectively closing the gate and leaving excess cholesterol to circulate freely in the blood.
The research team, led by Professors Carles J. Ciudad and Verònica Noé of the University of Barcelona, designed PPRHs to intercept this process before it even begins. By targeting the gene responsible for creating PCSK9, these molecules prevent the protein from being synthesized. When PCSK9 levels drop, the liver’s LDL receptor count increases, allowing the body to naturally and efficiently clear cholesterol from the blood.
A Chronology of Discovery: From Bench to Biological Model
The journey toward this innovation began with the identification of specific genetic sequences within the PCSK9 gene that were vulnerable to interference. The research team focused on two specific sequences located in exons 9 and 12 of the gene.
Early Stage: The Design of HpE9 and HpE12
The researchers engineered two unique polypurine hairpins, designated as HpE9 and HpE12. These are short, synthetic strands of DNA designed to bind with extreme specificity to the target genetic code. Through a process governed by Watson-Crick base pairing, these hairpins anchor themselves to the DNA/RNA, effectively acting as a "molecular block."
In Vitro Validation
The initial testing phase utilized HepG2 cells—a standard human liver cell line used in metabolic research. The results were striking:
- HpE12 performance: The therapy achieved a 74% reduction in PCSK9 RNA and an 87% reduction in the corresponding protein levels.
- Precision: The interaction between the PPRH and the target exon was highly stable, proving that the mechanism was not merely accidental but highly targeted.
In Vivo Success
Moving from the petri dish to living systems, the team utilized transgenic mice engineered to express human PCSK9. A single injection of the HpE12 molecule yielded rapid results. Within just 72 hours, the mice exhibited a 50% decrease in plasma PCSK9 and, crucially, a 47% reduction in total cholesterol levels. This demonstrated that the therapy is not only effective at the cellular level but is also capable of creating a systemic metabolic shift in a complex mammalian organism.
Supporting Data: Why PPRHs Outperform Current Standards
The medical community has been moving toward PCSK9 inhibition for years, with treatments like evolocumab, alirocumab, and the siRNA-based drug Inclisiran already available. However, the University of Barcelona’s approach brings several distinct advantages to the table.
Stability and Immunogenicity
Many biological therapies, such as monoclonal antibodies, are susceptible to being recognized by the immune system as "foreign," which can lead to diminished efficacy over time. PPRHs, being composed of standard DNA, are generally non-immunogenic. Furthermore, their structural stability makes them more resilient to degradation within the bloodstream, potentially allowing for less frequent dosing schedules.
Cost-Efficiency
One of the primary barriers to widespread adoption of current biological therapies is the exorbitant cost of manufacturing monoclonal antibodies. Synthesizing DNA-based oligonucleotides is significantly more cost-effective and scalable, which could democratize access to advanced cardiovascular care in lower-income settings or for patients without comprehensive insurance coverage.
Avoiding the "Statin Trap"
Perhaps the most significant clinical implication involves the avoidance of side effects. Statins, while effective, are notorious for causing muscle pain, fatigue, and, in some cases, increasing the risk of Type 2 diabetes. By focusing on the genetic root cause of LDL receptor degradation rather than the cholesterol-production pathway (the HMG-CoA reductase pathway targeted by statins), the PPRH approach avoids the systemic metabolic disruption that leads to statin-related myopathy.
Official Responses: Insights from the Lead Researchers
The study represents a massive interdisciplinary effort, involving the Faculty of Pharmacy and Food Sciences at the University of Barcelona, the Institute of Nanoscience and Nanotechnology (IN2UB), and the expertise of Nathalie Pamir at the University of Oregon.
Professor Carles J. Ciudad provided technical insight into the binding mechanism: "Specifically, one of the arms of each chain of the HpE9 and HpE12 polypurines binds specifically to polypyrimidine sequences of exons 9 and 12 of PCSK9, respectively, via Watson-Crick bonds. This interaction blocks gene transcription and interferes with the activity of RNA polymerase or transcription factors."
Professor Verònica Noé highlighted the clinical significance of the findings, noting that the rapid response in the animal models indicates that this is a highly potent therapeutic candidate. "The results show that both HpE9 and HpE12 are highly effective in HepG2 cells… In the case of transgenic mice, a single injection of HpE12 reduces plasma PCSK9 levels by 50% and cholesterol levels by 47% on the third day."
The project, which received funding from the Spanish Ministry of Science, Innovation and Universities (MICINN) and the National Institutes of Health (NIH) in the United States, stands as a testament to the power of international research cooperation in solving global health crises.
Implications: The Future of Cardiovascular Medicine
The implications of this research extend far beyond a single laboratory success. As heart disease continues to be the leading cause of mortality globally, the demand for "precision medicine" in cardiology has never been higher.
The Shift Toward Genetic Medicine
This study marks a significant milestone in the broader shift toward oligonucleotide therapeutics. By proving that DNA hairpins can reliably and safely downregulate problematic proteins, the research team has opened the door for similar PPRH-based treatments for other conditions involving protein overexpression, such as certain cancers or inflammatory disorders.
Clinical Roadmap
While the results in mice are encouraging, the research team acknowledges that the path to the pharmacy shelf requires rigorous clinical trials in humans. Future studies will need to focus on:
- Pharmacokinetics: Determining the exact duration of the cholesterol-lowering effect after a single dose.
- Long-term Safety: Ensuring that there are no "off-target" effects—meaning the hairpins do not accidentally bind to other genes and disrupt healthy cellular function.
- Delivery Mechanisms: Developing the most efficient way to ensure the PPRHs reach the liver cells (the primary site of PCSK9 production) without being cleared by the kidneys too quickly.
A New Era of Prevention
If these findings are validated in human clinical trials, we may be looking at the next generation of cardiovascular preventative care. Instead of a daily pill that affects the entire body’s metabolism, patients might eventually receive periodic, highly specific genetic injections that "fine-tune" their liver to clear cholesterol more efficiently.
For the millions of patients who struggle to maintain healthy cholesterol levels despite lifestyle changes and existing medication, the work of the University of Barcelona and the University of Oregon offers more than just hope—it offers a scientifically sound, targeted, and potentially life-saving future. By intercepting the disease at the genetic level, we are moving away from treating the symptoms of plaque buildup and toward managing the biological instructions that cause the problem in the first place.