Asthma remains one of the most pervasive chronic health challenges for children in the United States, affecting approximately 4.5 million young patients. For parents and clinicians alike, the condition has historically been managed through a reactive model: waiting for symptoms—such as wheezing, chest tightness, or labored breathing—to manifest before seeking clinical intervention. However, a groundbreaking partnership between Cedars-Sinai Guerin Children’s and Singapore-based health-tech firm Aevice Health is signaling a paradigm shift toward a proactive, data-driven future. By introducing the AeviceMD, a coin-sized wearable stethoscope, the medical community is moving closer to an era where respiratory distress is caught, assessed, and treated long before a child requires an emergency room visit.
The Core Innovation: A New Standard for Monitoring
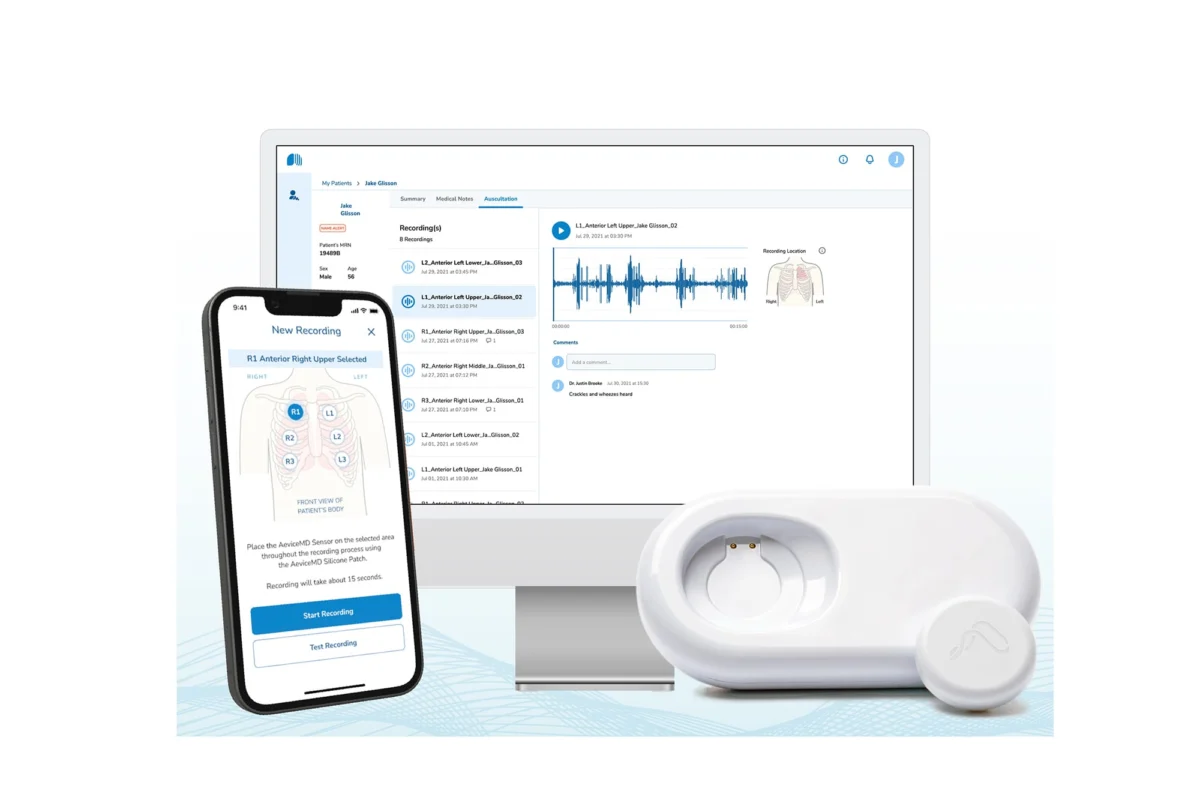
The AeviceMD device represents a significant leap forward in remote patient monitoring (RPM). Measuring roughly the diameter of a half-dollar coin, this lightweight, wearable sensor adheres to a child’s chest, allowing for continuous or on-demand monitoring of lung sounds for up to 10 hours at a time. Designed specifically for pediatric patients aged three and older, the device eliminates the ambiguity often associated with "parental observation," which, while well-intentioned, can be subjective and unreliable during the high-stress moments of an asthma flare-up.
By capturing real-time acoustic data, the device provides clinicians with a digital "map" of a child’s respiratory health. This data is transmitted securely, allowing doctors to track how a patient’s lungs sound overnight, during physical activity, or during periods of suspected illness. This longitudinal data stream is transformative; it allows physicians to move away from snapshots of health taken during rare clinic visits and toward a comprehensive, continuous understanding of how a child’s asthma evolves over weeks and months.
A Chronology of Collaboration and Development
The journey of the AeviceMD from a concept to a clinical reality is a testament to the power of international medical partnerships.
- 2022: The collaboration between Cedars-Sinai and Aevice Health officially began. The partnership was rooted in a shared goal: to solve the "blind spot" in pediatric asthma management. Physicians at Cedars-Sinai Guerin Children’s provided critical clinical insights, ensuring the device’s interface and data output met the rigorous standards of pediatric care.
- 2022–2023: A multi-phase pilot program was initiated. During this period, clinicians evaluated the device’s accuracy in detecting abnormal lung sounds, such as crackles or wheezing, against traditional auscultation methods.
- 2023: The culmination of these efforts arrived when the AeviceMD received official clearance from the U.S. Food and Drug Administration (FDA). This regulatory milestone cleared the path for the current rollout at Cedars-Sinai.
- 2024: Following the successful pilot and regulatory approval, Cedars-Sinai Guerin Children’s officially integrated the device into its pediatric asthma management program, marking the beginning of a new clinical standard for their patients.
Supporting Data and the Burden of Asthma
To understand the necessity of this technology, one must examine the current landscape of pediatric asthma. Asthma is not merely a common diagnosis; it is a leading cause of school absenteeism and emergency department utilization.
The Reliability Gap
Traditionally, pediatricians have relied on a "symptom-diary" approach. Caregivers are asked to track their child’s coughing frequency and perceived difficulty breathing. However, studies have repeatedly shown that parental perception of asthma control can fluctuate based on education levels, anxiety, and the time of day. Nocturnal asthma—symptoms that occur while the child is sleeping—is particularly dangerous, as it often goes undetected until the child is severely distressed.
The AeviceMD Performance
The AeviceMD addresses this by digitizing the "gold standard" of respiratory assessment: the physical exam. By providing a recorded, audible history of lung sounds, the device offers objective data. Clinicians can listen to the recordings remotely, compare them to previous sessions, and assess the efficacy of prescribed medications. If a child is on a maintenance inhaler, the device can quantify whether that medication is effectively reducing the frequency of adventitious breath sounds, providing a metric-based approach to treatment adjustments.
Official Perspectives: The Clinical and Executive Vision
The successful implementation of this program is the result of a concerted effort by both medical practitioners and administrative leaders who view technology as the ultimate force multiplier for patient care.
The Clinical Perspective: Dr. Irina Dralyuk
Dr. Irina Dralyuk, a pediatric pulmonologist at Guerin Children’s, has been at the forefront of this initiative. In a recent statement, she emphasized the importance of visibility. "I saw strong potential in this technology to support how we monitor and manage children with reactive airway issues like asthma outside of the clinical setting," Dr. Dralyuk noted.
She highlights that the most significant benefit is the timing of intervention. "Being able to assess respiratory symptoms in real time, especially overnight, provides valuable information that can guide earlier intervention and help prevent more serious breathing complications." For Dr. Dralyuk, the ultimate success metric is the reduction of systemic health burdens: "What we’re trying to do is prevent exacerbations, reduce hospitalizations and emergency department visits, and limit the need for interventions such as systemic steroid use to control asthma symptoms."
The Strategic Perspective: Adrian Ang and Nirdesh K. Gupta
From the development side, Adrian Ang, CEO of Aevice Health, views the partnership as a validation of his company’s core philosophy. "We built AeviceMD to make this kind of continuous symptom visibility possible," said Ang. "Having our technology deployed at a large, world-class healthcare system like Cedars-Sinai validates our belief that there is a critical, unmet patient need."
Nirdesh K. Gupta, PhD, managing partner of Cedars-Sinai Intellectual Property Company, underscored the importance of the institutional support provided to startups. He noted that the collaboration highlights how Cedars-Sinai facilitates the translation of innovation into daily clinical practice. "It’s an exciting example of how innovation can directly impact care and improve the way we manage chronic conditions like asthma," Gupta said.
Implications for the Future of Healthcare
The integration of the AeviceMD into the Cedars-Sinai ecosystem serves as a template for the future of chronic disease management. There are three primary implications for the broader medical community:
1. The Shift to "Hospital-at-Home"
The AeviceMD is part of a larger trend toward moving medical-grade monitoring out of the hospital and into the home. By decentralizing care, institutions can provide higher-quality monitoring at a fraction of the cost of an in-patient stay. This shift is particularly vital for pediatric populations, where the hospital environment can be distressing and disruptive to family life.
2. Precision Medicine in Asthma
Currently, asthma treatment is often "trial and error." A physician prescribes a controller medication and waits for the patient to report back in three months. With tools like the AeviceMD, medicine becomes more precise. Clinicians can observe the physiological response to a medication change within days, allowing for faster titration and a more tailored approach to the individual patient’s biology.
3. Reducing Healthcare Disparities
While technology access is often a concern, the simplicity and portability of the AeviceMD offer a pathway to reducing disparities in care. For families who live far from major medical centers or who struggle with the logistics of frequent clinic visits, remote monitoring acts as a digital lifeline. It ensures that regardless of geography, the child’s lung health remains under the expert supervision of specialists at centers like Cedars-Sinai.
Conclusion: A Proactive Path Forward
The partnership between Cedars-Sinai Guerin Children’s and Aevice Health is more than just a procurement of new technology; it is a fundamental shift in the philosophy of care. By empowering parents with tools to monitor their children and equipping physicians with the data necessary to act before a crisis occurs, the healthcare industry is finally catching up to the needs of chronic asthma patients.
As the program continues to grow and collect data, the findings will likely influence national standards for pediatric asthma management. The goal is clear: to ensure that "breathing easier" is not just a hope for children with asthma, but a measurable, achievable reality through the convergence of clinical expertise and digital innovation. In the coming years, we can expect to see more such wearables entering the clinical space, further reducing the reliance on emergency interventions and giving millions of children the freedom to live active, unburdened lives.