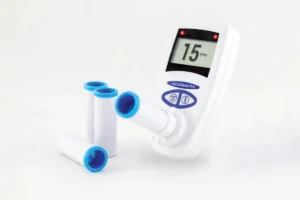
Washington D.C. – [Current Date] – The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to the Encora X1, a pioneering noninvasive, wrist-worn neurostimulation device developed by Encora Therapeutics. This landmark decision ushers in a new era of treatment options for adults living with essential tremor (ET), a neurological disorder characterized by involuntary, rhythmic shaking, primarily affecting the hands. The Encora X1 is designed to significantly reduce hand tremors and enhance daily functional abilities, offering a much-needed alternative to conventional pharmacological and invasive surgical interventions.
Main Facts: A New Era for Essential Tremor Management
The clearance of the Encora X1 represents a pivotal moment for millions grappling with the debilitating effects of essential tremor. This novel device promises to restore a degree of independence and quality of life previously elusive for many patients.
Breakthrough Device Receives FDA Clearance
Encora Therapeutics, a company at the forefront of non-invasive neuromodulation technology, officially announced the FDA’s 510(k) clearance for its flagship product, the Encora X1. This regulatory approval signifies that the device has been found substantially equivalent to existing legally marketed predicate devices, affirming its safety and efficacy for its intended use. Unlike many existing treatments that involve systemic medication or complex surgical procedures, the Encora X1 offers a discreet, wearable solution that patients can integrate seamlessly into their daily routines. It operates on a prescription-only basis, ensuring that its application is guided by professional medical oversight.
Addressing a Significant Unmet Need
Essential tremor is one of the most common movement disorders globally, affecting an estimated 7 million Americans. Its prevalence often surpasses that of Parkinson’s disease, yet public awareness and effective, non-invasive treatment options have historically lagged. Patients with ET frequently experience profound difficulties with fundamental daily activities such as eating, drinking, writing, dressing, and performing fine motor tasks. This often leads to significant social anxiety, professional limitations, and a diminished sense of self-sufficiency. Current treatment paradigms typically involve medications that can have systemic side effects or, for severe cases, invasive deep brain stimulation (DBS) surgery, which carries inherent risks and is not suitable for all patients. The Encora X1 directly addresses this critical gap, providing a non-pharmacological, non-surgical alternative for managing symptoms.
The Technology Behind the Innovation
At the heart of the Encora X1’s functionality lies sophisticated proprietary algorithms that enable real-time analysis of a patient’s tremor patterns. The device, roughly the size and appearance of a modern smartwatch, is worn on the wrist. Once activated, it delivers targeted electrical stimulation to the neural pathways responsible for generating the tremor. This customized stimulation is designed to disrupt the errant tremor signals, thereby stabilizing the limb and improving motor control. The device’s lightweight and discreet design are crucial elements, empowering patients to manage their symptoms on demand and with minimal disruption to their personal and professional lives. This "on-the-go" relief represents a significant advancement over treatments requiring fixed schedules or clinical visits for adjustment.
Chronology: From Concept to Clinical Reality
The journey of the Encora X1 from an innovative concept to an FDA-cleared medical device is a testament to rigorous scientific inquiry, dedicated research, and a patient-centric development philosophy.
The Genesis of Encora Therapeutics
Encora Therapeutics was founded with a clear and ambitious mission: to restore independence for individuals suffering from movement disorders. Recognizing the limitations of existing treatments for essential tremor, the company embarked on developing non-invasive neuromodulation technologies. Their approach centered on leveraging advanced signal processing and targeted stimulation to address the neurological underpinnings of tremor without the need for surgery or systemic medication. This foundational vision guided every stage of the Encora X1’s development, from initial research and design to clinical trials and regulatory engagement. The co-founders and leadership team brought together expertise in neuroscience, biomedical engineering, and business operations to translate this vision into a tangible solution.
Rigorous Clinical Validation
Before receiving FDA clearance, the Encora X1 underwent a comprehensive clinical evaluation designed to establish its safety and effectiveness. The regulatory submission was bolstered by compelling data derived from two pivotal clinical studies. The first was a randomized, sham-controlled trial, considered the gold standard for evaluating medical interventions. In this type of study, participants are randomly assigned to receive either the active device or a placebo (sham) device, allowing researchers to isolate the true effect of the intervention while minimizing bias. The second was a 90-day home-use study, which provided invaluable insights into the device’s performance and patient experience in real-world, daily living scenarios. These studies were instrumental in demonstrating consistent improvements in both objective measures of tremor severity and subjective assessments of functional tasks critical to daily life.
The FDA’s Pathway to Clearance
The FDA 510(k) clearance pathway is designed for medical devices that are substantially equivalent to a legally marketed predicate device, meaning they have the same intended use and technological characteristics, or have different technological characteristics but do not raise new questions of safety and effectiveness. Encora Therapeutics meticulously compiled and submitted extensive data from their clinical trials, alongside detailed information on the device’s design, manufacturing, and safety profile. The FDA’s rigorous review process ensures that new medical devices meet stringent standards for patient protection and performance. The successful navigation of this process by Encora Therapeutics underscores the robust scientific evidence supporting the Encora X1’s claims and its readiness for commercial marketing. This clearance specifically authorizes the device as a prescription wearable to aid in the relief of hand tremors in the treated limb during stimulation in adults with essential tremor.
Supporting Data: Evidence of Efficacy and Safety
The foundation of the Encora X1’s FDA clearance rests firmly on the robust clinical data demonstrating its ability to alleviate tremors and improve patients’ functional independence.
Insights from Key Clinical Trials
The two pivotal clinical studies conducted by Encora Therapeutics provided the necessary evidence for regulatory approval. The randomized, sham-controlled trial, with its rigorous methodology, showed statistically significant improvements in tremor severity among participants using the active Encora X1 device compared to those using the sham device. This type of controlled environment is crucial for proving causality—that the device, and not external factors or the placebo effect, was responsible for the observed benefits. The subsequent 90-day home-use study further corroborated these findings, providing long-term data on the device’s sustained efficacy and usability in the naturalistic setting of patients’ homes. This real-world evidence is vital for understanding how a device performs outside of a controlled clinical environment, offering a more complete picture of its practical utility.
Quantifying Tremor Reduction and Functional Improvement
The clinical data consistently highlighted the Encora X1’s capacity to reduce tremor severity, which directly translated into measurable improvements in the ability to perform daily functional tasks. Patients reported and demonstrated enhanced capabilities in activities that are often severely impacted by ET, such as:
- Eating: Holding utensils steady, bringing food to the mouth without spilling.
- Drinking: Lifting a cup or glass to the lips without spillage.
- Writing: Producing legible handwriting, a common challenge for ET patients.
- Fine Motor Skills: Tasks like dialing a telephone, plugging a cord into an electric outlet, buttoning clothes, or applying makeup.
These improvements are not merely cosmetic; they directly impact a patient’s ability to live more independently, participate in social activities, and maintain a sense of dignity and control over their lives. The objective measures of tremor reduction, combined with the subjective reports of improved functional outcomes, paint a comprehensive picture of the Encora X1’s therapeutic potential.
A Favorable Safety Profile
A critical aspect of any medical device evaluation is its safety profile. In the clinical trials for the Encora X1, device-related adverse events were consistently reported as non-serious and self-resolving. This indicates that while side effects may occur, they are generally mild and temporary, not requiring significant medical intervention. The reported side effects included:
- Numbness in the treated limb.
- Skin irritation or rash at the application site.
- Temporary muscle weakness.
- Localized discomfort or pain.
- A buzzing or tingling sensation during stimulation.
- In some instances, a possible temporary worsening of tremor.
The transient nature and mild severity of these adverse events contribute to a favorable risk-benefit profile, especially when weighed against the potential benefits of tremor reduction and improved functional independence. As a prescription-only device, its use will be guided by healthcare professionals who can assess individual patient suitability and manage any potential side effects.
Official Responses: Voices from the Medical and Corporate Spheres
The FDA clearance has been met with significant enthusiasm from both the medical community and the leadership of Encora Therapeutics, reflecting the anticipation for this novel treatment option.
Medical Experts Hailing the Advance
Dr. Ann Murray, a principal investigator in the Encora X1 trials and Chief of the WVU Rockefeller Neuroscience Institute (RNI) Comprehensive Movement Disorder Center, articulated the profound impact of this innovation. "The data demonstrate meaningful improvement in tremor severity and functional outcomes for patients with limited options," Dr. Murray stated. Her commentary underscores the current treatment dilemma for ET patients: "For the 7 million Americans suffering from essential tremor, medications are often ineffective or poorly tolerated, leaving invasive brain therapy as the only alternative." Dr. Murray emphasized the significance of non-invasive technologies like the Encora X1, calling them "an important area of innovation in the treatment of essential tremor." Her perspective highlights the medical community’s eagerness for effective, less intrusive solutions that can bridge the gap between medication failure and the necessity of surgery.
Encora Therapeutics Leadership on Vision and Impact
The leadership at Encora Therapeutics expressed deep satisfaction and renewed commitment following the FDA clearance. Alli Davanzo, co-founder and vice president of business operations, shared the foundational ethos of the company: "We started Encora with a simple goal: to restore independence for movement disorder patients. Today, we are one step closer to making that vision a reality." Davanzo emphasized the empowering nature of the Encora X1, stating, "Encora X1 offers a non-surgical, drug-free alternative, allowing patients to manage their symptoms on their own terms." This speaks to the autonomy and control the device offers patients, a crucial factor in improving quality of life.
Nadim Yared, Chief Executive Officer of Encora Therapeutics, echoed this sentiment, underscoring the diligent effort behind the device’s development. "This clearance represents the culmination of rigorous research and patient-centric design," Yared remarked. He conveyed the company’s eagerness to bring this innovation to market: "We are eager to bring this novel solution to patients who have long waited for more options." These statements collectively highlight Encora’s commitment to innovation, patient well-being, and addressing critical unmet needs within the neurological disorder landscape.
Patient Advocates’ Perspective (Inferred)
While specific patient advocates were not quoted in the original release, the implications of such a device are clear for the patient community. Organizations dedicated to essential tremor awareness and support are likely to welcome the Encora X1 as a beacon of hope. For years, patients have navigated a challenging path, often feeling misunderstood or underserved by existing therapies. The introduction of a non-invasive, drug-free option provides a renewed sense of possibility for regaining control over daily activities and improving overall quality of life. Patient advocacy groups would likely emphasize the importance of accessibility, education for both patients and healthcare providers, and continued research into personalized treatment approaches for ET. The ability to manage symptoms "on their own terms," as articulated by Encora’s co-founder, resonates deeply with patients striving for autonomy and a reduction in the psychosocial burden of living with a visible movement disorder.
Implications: Reshaping the Landscape of Essential Tremor Treatment
The FDA clearance of the Encora X1 is poised to have far-reaching implications, fundamentally altering how essential tremor is managed and perceived, both clinically and socially.
A Paradigm Shift for Patients and Clinicians
For patients, the Encora X1 offers a significant expansion of treatment choices, moving beyond the traditional binary of medication or surgery. It provides a non-invasive, on-demand solution that can be tailored to individual needs, offering a level of flexibility and control previously unavailable. This could lead to earlier intervention for those who find medications intolerable or ineffective, potentially delaying or even obviating the need for more invasive procedures. For clinicians, the device presents a valuable tool in their therapeutic arsenal. It allows for a more stratified approach to ET management, enabling them to offer a less aggressive intervention before considering surgery, and providing an option for patients who are not candidates for or decline surgical options. The ability to prescribe a wearable device that directly addresses a patient’s functional needs during daily activities empowers physicians to offer more personalized and patient-centered care.
Economic and Quality of Life Benefits
The economic implications of the Encora X1 could be substantial. By offering an effective non-surgical alternative, it may reduce the overall healthcare burden associated with essential tremor, potentially lowering the costs related to repeated medication trials, managing side effects, or complex surgical procedures and post-operative care. More importantly, the improvements in functional independence can have profound quality of life benefits. Patients who can eat, drink, write, and perform self-care tasks with greater ease are likely to experience reduced social anxiety, improved self-esteem, and increased participation in professional and recreational activities. This return to normalcy can enhance mental well-being and overall life satisfaction, contributing to a more engaged and productive societal contribution. The ability to manage tremors discreetly and effectively can also help mitigate the social stigma often associated with visible movement disorders.
The Future of Neuromodulation and Wearable Technology
The success of the Encora X1 heralds a promising future for non-invasive neuromodulation and wearable medical technology. This device underscores the growing trend towards personalized, on-demand therapies that leverage smart technology to address chronic conditions. It demonstrates the potential for intelligent wearables to monitor physiological signals, deliver targeted interventions, and provide real-time relief, thereby shifting the paradigm of healthcare from reactive to proactive and preventative. The integration of proprietary algorithms with discreet form factors opens doors for similar innovations in managing other neurological conditions, pain management, and rehabilitation. It could inspire further research into the precise neural pathways involved in various movement disorders and lead to even more sophisticated and individualized therapeutic devices. The Encora X1 is not just a device for essential tremor; it is a blueprint for the next generation of smart health solutions.
Important Safety Considerations and Accessibility
While the Encora X1 offers significant promise, it is crucial to reiterate the important safety information provided by Encora Therapeutics. The device is available by prescription only, emphasizing the necessity of a physician’s evaluation to determine suitability and to monitor its use. It is explicitly stated that the Encora X1 is not a cure for essential tremor, but rather an aid for symptom relief during stimulation. Patients and healthcare providers must be aware of the potential, though generally mild and self-resolving, side effects. Federal law restricts the sale of this device to prescription by or on the order of a physician, reinforcing the need for medical guidance.
As Encora Therapeutics moves towards commercialization, critical questions of accessibility, insurance coverage, and patient education will emerge. Ensuring that this innovative solution reaches the broadest possible population of essential tremor sufferers will be paramount, requiring collaboration among manufacturers, healthcare providers, insurers, and patient advocacy groups. The Encora X1 represents a significant leap forward, offering renewed hope and practical solutions for millions seeking to regain control over their lives in the face of essential tremor.